Staphylococcus aureus small colony variants show common metabolic features in central metabolism irrespective of the underlying auxotrophism
- 1Institute of Medical Microbiology, University of Münster, Münster, Germany
- 2Lehrstuhl für Biochemie, Technische Universität München, Garching, Germany
- 3Department of Microbiology and Immunology, University of Wisconsin School of Medicine and Public Health, Madison, WI, USA
In addition to the classical phenotype, Staphylococcus aureus may exhibit the small colony-variant (SCV) phenotype, which has been associated with chronic, persistent and/or relapsing infections. SCVs are characterized by common phenotypic features such as slow growth, altered susceptibility to antibiotic agents and pathogenic traits based on increased internalization and intracellular persistence. They show frequently auxotrophies mainly based on two different mechanisms: (i) deficiencies in electron transport as shown for menadione- and/or hemin-auxotrophs and (ii) thymidylate biosynthetic-defective SCVs. To get a comprehensive overview of the metabolic differences between both phenotypes, we compared sets of clinically derived menadione-, hemin- and thymidine-auxotrophic SCVs and stable site directed mutants exhibiting the SCV phenotype with their corresponding isogenic parental strains displaying the normal phenotype. Isotopologue profiling and transcriptional analysis of central genes involved in carbon metabolism, revealed large differences between both phenotypes. Labeling experiments with [U-13C6]glucose showed reduced 13C incorporation into aspartate and glutamate from all SCVs irrespective of the underlying auxotrophism. More specifically, these SCVs showed decreased fractions of 13C2-aspartate and glutamate; 13C3-glutamate was not detected at all in the SCVs. In comparison to the patterns in the corresponding experiment with the classical S. aureus phenotype, this indicated a reduced carbon flux via the citric acid cycle in all SCV phenotypes. Indeed, the aconitase-encoding gene (acnA) was found down-regulated in all SCV phenotypes under study. In conclusion, all SCV phenotypes including clinical isolates and site-directed mutants displaying the SCV phenotype were characterized by down-regulation of citric acid cycle activity. The common metabolic features in central carbon metabolism found in all SCVs may explain similar characteristics of the S. aureus SCVs irrespective of their auxotrophism as well as the specific genetic and/or regulatory backgrounds.
Introduction
Staphylococcus aureus (S. aureus) has been recognized as one of the most important human pathogens world-wide causing a wide range of mild to serious infections within and outside the hospital aggravated by the dissemination of different methicillin-resistant S. aureus (MRSA) lineages (Lowy, 1998; David and Daum, 2010). Besides its capability to cause acute infections, S. aureus can cause chronic courses of infection despite adequate antimicrobial therapy that are often associated with a defined S. aureus phenotype, designated as small-colony variants (SCVs) (Proctor et al., 2006). SCVs represent a sub-population with distinct phenotypic and pathogenic traits adapted to an intracellular lifestyle (von Eiff et al., 2001, 2006; Sachse et al., 2010; Tuchscherr et al., 2010). As main feature, they show frequently auxotrophies (auxotrophism) for menadione, hemin and/or thymidine, however, strains without any detectable auxotrophy or with other auxotrophies including those for CO2 and thiamin have been described (Thomas, 1955; von Eiff et al., 1997; Kahl et al., 2003; Chatterjee et al., 2008; Lannergård et al., 2008; Gómez-González et al., 2010). The best-investigated and most prevalent SCV phenotypes, the menadione and/or hemin autotrophic SCVs as well as thymidine autotrophic SCVs, are characterized by deficiencies in the electron transport and in the thymidylate biosynthetic pathway, respectively (von Eiff et al., 1997; Chatterjee et al., 2008). It has been shown for hemin and menadione auxotrophic SCVs, based on mutations in hemB and menD (von Eiff et al., 1997; Kohler et al., 2003, 2008), that genes involved in the central metabolic processes were affected. Transcriptomic and proteomic approaches revealed considerable differences between the wild type and SCV phenotypes especially in the fermentative pathways (Kohler et al., 2003, 2008; Seggewiss et al., 2006). However, because clinically derived SCVs tend to revert quickly back into the wild type phenotype, most of SCV studies were performed with genetically defined, stable mutants. A recent proteomic study comparing a clinically derived SCV with a corresponding hemB mutant SCV and a gentamicin-induced SCV revealed common, but also distinct features between naturally occurring and genetically generated SCVs apart from changes triggered by the mutational inactivation of the electron transport chain (Kriegeskorte et al., 2011). Nevertheless, the complex metabolic and physiological changes along with the SCV phenotype are still not fully understood and more multifaceted than revealed from studies with genetically defined mutants displaying the SCV phenotype.
The aim of this study was to get additional insights into the metabolic properties of S. aureus SCVs as compared to their corresponding isogenic normal phenotype. For this purpose, we investigated a comprehensive set of SCVs including both clinically derived strains and stable site directed mutants by 13C-isotopologue profiling and transcriptional analysis.
Materials and Methods
Bacterial Strains and Culture Conditions
Clinical S. aureus wild types and SCVs were recovered in parallel from patients with chronic infections (e.g., osteomyelitis and cystic fibrosis). Clonality was verified by SmaI macrorestriction analyses by pulsed-field gel electrophoresis (PFGE). Strains used in this study were summarized in Table 1. The S. aureus isolates were grown on Columbia sheep blood agar and tryptic soy agar at 37°C for 24–48 h. Liquid cultures were grown aerobically in 50 ml tryptic soy broth (TSB) in 500 ml flasks at 37°C and 160 rpm. For labeling experiments (isotopolog profiling) TSB without dextrose (Bacto Tryptic Soy without dextrose, BD, New Jersey, USA) including 17 g of pancreatic digest of casein, 3 g of enzymatic digest of sojabean meal, 5 g of sodium chloride and 2.5 g of dipotassium phosphate was used. The medium was supplemented with 2.5 g of [U-13C6]glucose.
Cell Isolation and Growth Curve Analysis
For isotopolog profiling, 50 ml cultures were inoculated to an optical density of 0.05 (578 nm) from overnight cultures. Cells were harvested after 540 min by centrifugation (10 min, 5000 × g, 4°C) and washed three times with 10 ml PBS. Pellets were stored at −80°C. Cells were resuspended in 10 ml PBS and autoclaved (20 min, 121°C). For the growth curve analysis, cultures were grown in 50 ml TSB in 500 ml flasks at 37°C on a rotary shaker at 160 rpm. The optical density was measured every hour at 578 nm using Ultraspec 1100 pro spectrophotometer (Amersham Biosciences, Freiburg, Germany).
Construction of a ΔhemB-Mutant in S. aureus 8325-4
The hemB knockout mutant of S. aureus 8325-4 was constructed by allelic replacement of the hemB gene with ermB cassette (mediating erythromycin resistance) using the vector pCE8 as described before (von Eiff et al., 1997). The mutant was verified by restriction analysis and sequencing.
Isotopologue Profiling
Bacterial cells (approximately 5 mg) were suspended in 0.5 ml of 6 M hydrochloric acid and incubated at 105°C for 24 h. The amino acids were purified on a Dowex 50W×8 column (washing 2 × 750 μl H20; developing 1 ml 2 M ammonium hydroxide). The eluate was dried under a steam of nitrogen and resuspended in 50 μl dry acetonitrile. 50 μl of N-(tert-butyldimethylsilyl)-N-methyl-trifluoroacetamide containing 1 % of tert-butyldimethylsilylchlorid were added and the mixture was incubated at 70°C for 30 min. The tert-butyl-dimethylsilyl derivatives of amino acids were then used for gas chromatography–mass spectrometry (GC/MS) and isotopolog analysis as described elsewhere (Eylert et al., 2008).
Semi-Quantitative RT-PCR
Total RNA was extracted from bacteria grown in TSB medium to late exponential growth phase using QuantiTect reverse transcription kit (QIAGEN) according to the manufacturer's recommendations. PCR reaction were performed with the CFX96 system (Bio-Rad Laboratories, München, Germany) under the following conditions: 95°C for 15 min, 50 cycles (95°C for 10 s, 10 s at 55°C for 10 s, 72°C for 30 s) using the EvaGreen Kit (Segentic, Borken, Germany). Three independent biological replicates were tested in duplicate. N-fold expression values relative to the house-keeping genes gyrB, gmk and aroE (normalized by the geometric mean of the relative quantities of all three reference genes) and for each strain set normalized to expression of the wild type isolate were calculated using CFX Manager v3.1 (Bio-Rad).
Results and Discussion
S. aureus SCVs show many common features, such as slow growth, reduced pigmentation and changed expression of virulence determinants, independent of their underlying auxotrophic phenotype, molecular mechanism of SCV phenotype generation or genetic strain background. A similar phenotypic appearance may reflect analogous metabolic properties or a similar metabolic status. To investigate the metabolic differences (i) between S. aureus normal and SCV phenotypes and (ii) between different kinds of SCVs by isotopolog profiling, respective isogenic strains sets were analyzed (Table 1) and regulatory differences in central metabolic and virulence related genes were determined.
Using a S. aureus strain “sextet,” consisting of three wild type isolates and three isolates displaying different SCV phenotypes including a clinically derived SCV, a site-directed hemB mutant and a gentamicin induced SCV, we identified significant phenotypic specific differences in the labeling patterns of amino acids (Figures 1A–C) (Kriegeskorte et al., 2011). In experiments with 2.5 g/l [U-13C6]glucose as a supplement to the TSB medium, all isolates, independent of their phenotype, showed high 13C incorporation of about 20–40% into alanin (Figures 1A, 7A), reflecting a high glycolytic activity in both phenotypes. As expected, under in vitro nutrient rich conditions, glucose served as the major energy source for growth of S. aureus. Nevertheless, the fraction of unlabeled amino acids in the experiment with [U-13C6]glucose reflected the pronounced capacity of S. aureus to uptake and to use external (unlabeled) amino acids or peptides from the medium. In comparison to alanine, more pronounced differences were noticed in the labeling patterns of glutamate and aspartate (Figures 1B,C). While all normal phenotypes showed a 13C-excess between 11.8 and 16.0% in aspartate and glutamate, all SCV phenotypes were characterized by a substantial reduced 13C-excess between 0.7 and 4.4% (Figure 1A). As glutamate and aspartate are directly linked to the citric acid cycle intermediates α-ketoglutarate and oxalacetate, respectively, via transamination, the reduced 13C-label of these amino acids could indicate a reduced citric acid cycle activity in all SCV phenotypes (Figure 5). The comparison of the averaged 13C-excess values between the three normal and the three SCV phenotypes revealed no significant differences in the 13C-excess of alanine, but a significant reduction of the 13C-excess of aspartate and glutamate in the SCV phenotypes (Figure 7A). Moreover, the isotopolog distributions in aspartate and glutamate from the SCVs were clearly different from the corresponding patterns in aspartate and glutamate from the wild-type phenotype and the revertant or complemented strains (Figures 1B,C). Whereas the later group was characterized by multiple 13C-isotopologs also comprising three and more 13C-atoms, the amino acids from the SCVs displayed higher fractions of 13C1-isotopologs. Again, this could reflect that the carbon flux via the citrate cycle producing oxaloacetate/Asp and α-ketoglutarate/Glu carrying multiple 13C-atoms in the experiments with [U-13C6]glucose is substantially reduced in the hemB mutant and the gentamicin induced SCVs. This hypothesis is in line with earlier conclusions made on the basis of proteome and transcriptome studies (Kohler et al., 2003, 2008; Seggewiss et al., 2006).
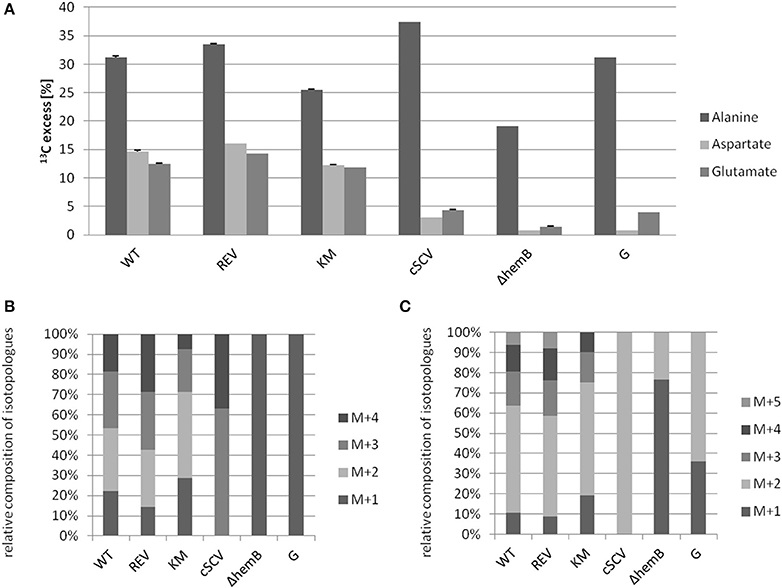
Figure 1. 13C-Excess in alanine, aspartate and glutamate from S. aureus strains grown with [U-13C6]glucose (A). Error bars indicate the standard deviation of three technical replicates. WT (clinically isolated normal phenotype), REV (spontanous revertant of the clinical SCV) and KM (complemented ΔhemB mutant with an intact hemB gene in trans) display an S. aureus wild-type phenotype. cSCV (clinically isolated SCV phenotype), ΔhemB (site-directed hemB mutant of WT) and G (gentamicin induced SCV phenotype) display a S. aureus SCV phenotype. Relative contribution of 13C-isotopologs carrying one to five 13C-atoms (M+1–M+5) in aspartate (B) and glutamate (C). The group of M+X comprises all isotopologs of aspartate and glutamate with only X labeled carbon units irrespective of the location within the molecule. Isotopologue patterns of amino acids with <1% 13C-excess are not shown due to the high standard deviations.
Interestingly, the clinical SCV showed similar patterns of the 13C-excess in aspartate and glutamate, in comparison to the profiles from the site-directed hemB mutant and to the gentamicin induced SCV, respectively (Figure 1A). However, the relative isotopolog distributions differed between the SCV phenotypes (Figures 1B,C). While in the hemB mutant and in the gentamicin induced SCV, the M+1 species were dominant, the 13C2-species were more abundant indicating that single runs, but no multiple runs, via the citric acid cycle were still operative in the clinically derived SCV. To investigate whether reduced carbon flux via the citrate cycle is a general feature of S. aureus SCVs that also includes the major auxotrophic phenotypes (hemin, menadione, and thymidine), we analyzed a comprehensive set of six isogenic strain sets, each comprising the normal wild type and different SCV phenotypes including clinical derived SCVs and genetically defined mutants displaying the SCV phenotype. The growth properties of the strain pairs are shown in Figures 2A–C, 7B. All SCVs showed a pronounced growth defect compared to their normal phenotypes, irrespective of the underlying auxotrophism and reached considerably lower optical densities under aerobic conditions. Again, 13C incorporation into alanine was highly efficient in all phenotypes and strains and no significant difference between the normal and the SCV phenotypes could be observed (Figures 3A–C, 7B). However, irrespective of the underlying auxotrophism, all SCV phenotypes again showed reduced 13C-incorporation into aspartate and glutamate, revealing the assumed reduced activity of carbon flux via the citric acid cycle (Figures 3A–C, 7B). This conclusion was verified by the isotopolog distribution as shown in Figures 4A–C. While all of the normal phenotypes showed in aspartate high fractions of the M+2 species, all SCVs were devoid of this species. In contrast, the SCV phenotypes showed increased relative fractions of M+1 and M+3 species indicating the reduced flux via the citric acid cycle with 13C2-isotopologs, but higher contributions of oxaloacetate/Asp formation by anaplerotic reactions [i.e., giving rise to the 13C3-isotopologs by carboxylation of [U-13C3]pyruvate or PEP in the SCVs (Figure 5)].
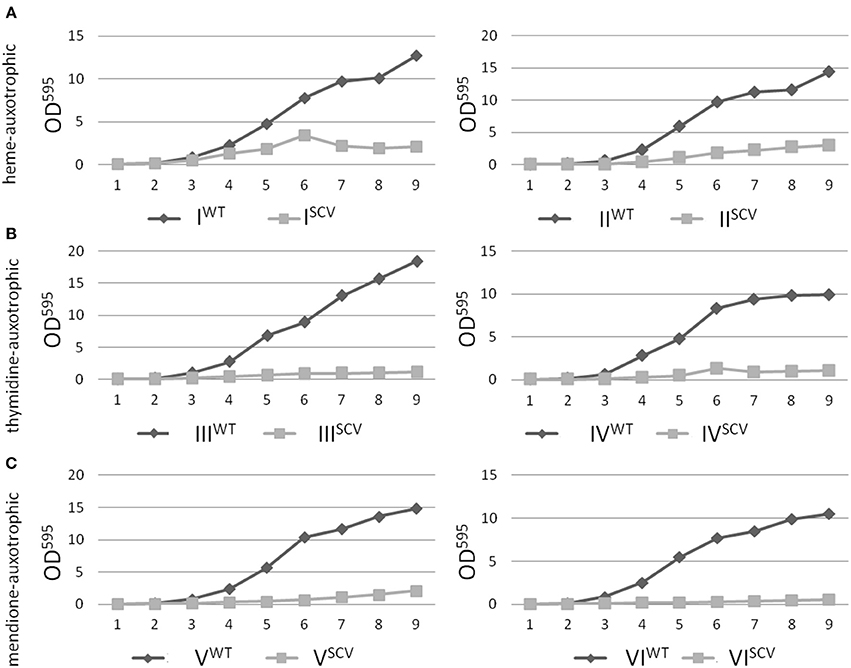
Figure 2. Growth curve analysis of S. aureus isolates with normal and SCV phenotype. Heme-auxotrophic (A), thymidine-auxotrophic (B) and menadione-auxotrophic (C). Strain pairs I, III, and V represent clinical isolates consisting of a normal phenotype (WT) and a SCV phenotype (SCV). Strain pairs II, IV, and VI consist of a normal phenotype and an in vitro generated SCV phenotype. The optical density of the cultures was measured at 595 nm and values were plotted against the time.
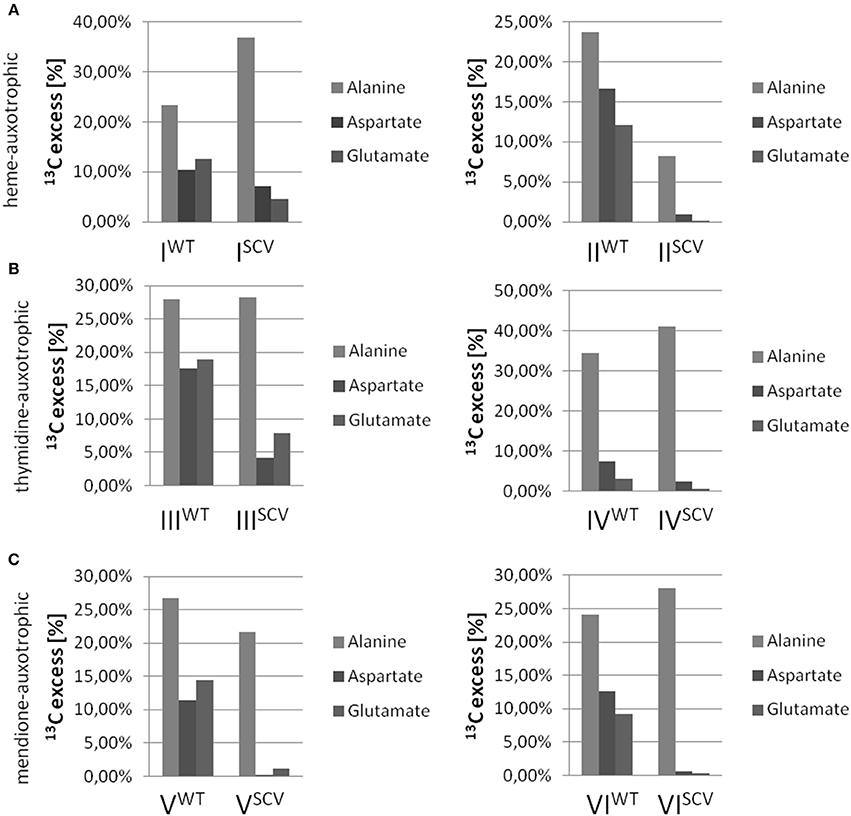
Figure 3. 13C-Excess in alanine, aspartate and glutamate from S. aureus strains grown with [U-13C6]glucose. Columns represent the mean values of three technical replicates; heme-auxotrophic (A), thymidine-auxotrophic (B) and menadione-auxotrophic (C). Strain pairs I, III, and V represent clinical isolates consisting of a normal phenotype (WT) and a SCV phenotype (SCV). Strain pairs II, IV, and VI consist of a normal phenotype and an in vitro generated SCV phenotype.
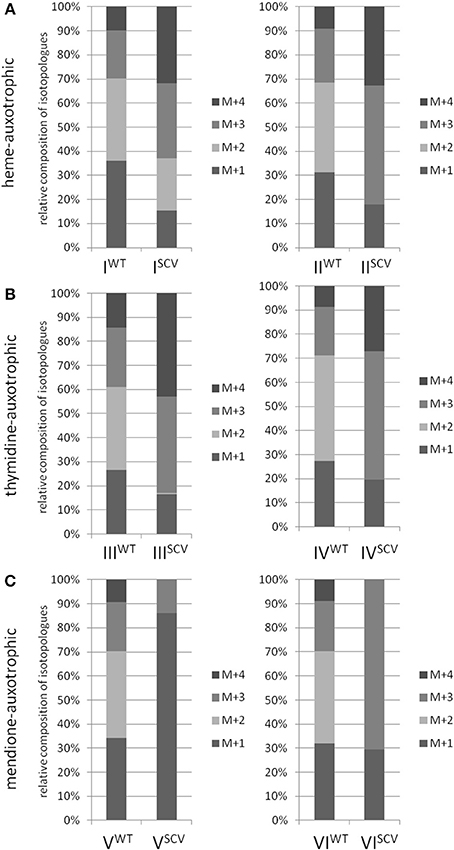
Figure 4. Isotopologue composition of aspartate. Comparison between normal and SCV phenotypes of S. aureus displaying different auxotrophisms. Heme-auxotrophic (A), thymidine-auxotrophic (B) and mendione-auxotrophic (C). Strain pairs I, III, and V represent clinical isolates. Strain pairs II, IV, and VI consist of a normal phenotype and an in vitro generated SCV phenotype. Patterned columns represent the mean values of three technical replicates. The relative contributions of each 13C-isotopolog (M+1–M+4) with regard to the overall enrichment were shown.
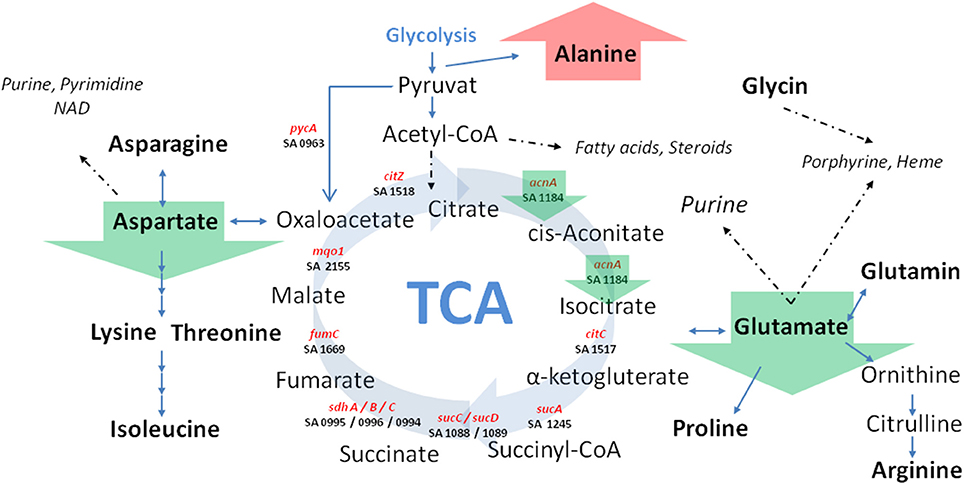
Figure 5. Schematic view of central carbon metabolism in S. aureus. Green arrows marked SCV specific differences irrespective of the underlying auxotrophism compared to their corresponding normal phenotypes. TCA: tricarboxylic acid cycle; Involved genes (marked in red) and corresponding KEGG gene identifier were shown.
Previously, we could show that the expression of acnA (aconitase) which catalyzes the first step of the TCA cycle (Figure 5), was reduced in a clinical SCV as well as in a site-directed hemB mutant (Seggewiss et al., 2006; Al Laham et al., 2007; Kriegeskorte et al., 2011). With the recent isotopolog data, down-regulation of acnA resulting in reduced carbon flux via the citric acid cycle seems to be a common feature of the SCV metabolism irrespective of the underlying molecular mechanism leading to this phenotype. Not surprisingly, all of the investigated clinical SCVs comprising heme, menadione and thymidine auxotrophic phenotypes, respectively, displayed a transcriptional down-regulation of acnA compared to their corresponding normal phenotype (Figures 6A–C, 7A,B). This is in line with previous studies on transcriptomic or proteomic level (Kohler et al., 2003; Seggewiss et al., 2006). Along with the decreased metabolic activity, all SCVs showed markedly reduced expression of the major virulence regulators hld and sigB. Hld is the effector molecule (a regulatory RNA) of the agr system which regulates virulence determinants in S. aureus such as the major toxins hla (α-hemolysin) and hlb (β-hemolysin). Corresponding to the reduced expression of hld, the investigated SCV phenotypes showed a clearly reduced hemolytic activity on Columbia blood agar plates (data not shown).
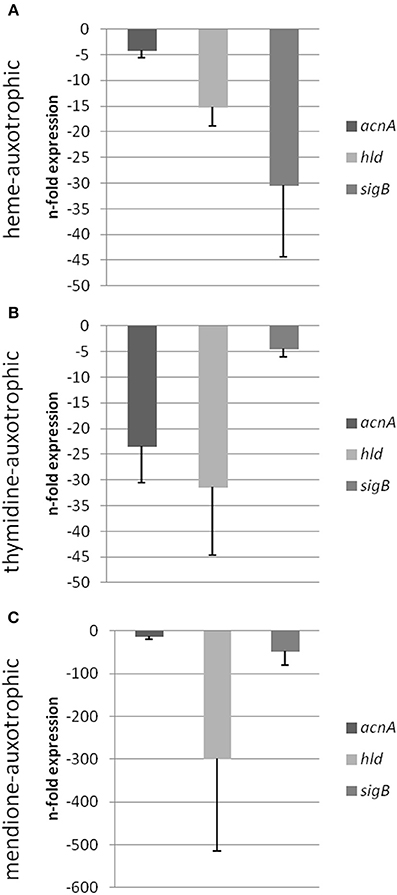
Figure 6. Semi-quantitative rt-PCR. Quantification of the expression of acnA, hld and sigB of different S. aureus strains displaying the SCV phenotype compared to its respective isogenic wild-type isolate. Expression data is normalized using three internal control genes (gmk, aroE, and gyrB) and displayed relative to the corresponding wild-type isolates (IWT, IIIWT, and VWT were set as 1). Isogenic strain pair IWT and ISCV [heme auxotrophic (A)], isogenic strain pair IIIWT and IIISCV [thymidine auxotrophic (B)] and isogenic strain pair VWT and VSCV [menadione auxotrophic (C)] were shown.
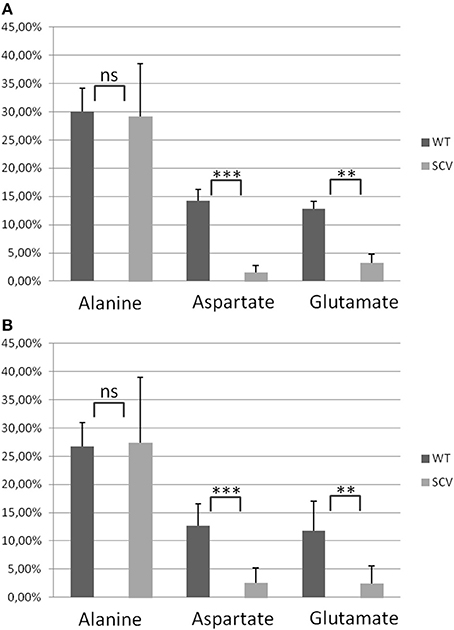
Figure 7. Averaged 13C-Excess in alanine, aspartate and glutamate from S. aureus normal and SCV phenotypes. (A) Columns represent the averaged values ± standard deviation (SD) of the normal phenotypes (WT) including S. aureus 3878 WT, REV and KM and the SCV phenotypes (SCV) including S. aureus cSCV, ΔhemB and G. (B) Columns represent the averaged values ± standard deviation (SD) of the normal phenotypes (WT) including S. aureus I-VIWT and the SCV phenotypes (SCV) including S. aureus I-VISCV. Statistical significance was assessed in pairwise comparison between WT and SCV for each amino acid with the two-tailed Student's t-test. **P < 0.01, ***P < 0.001.
In conclusion, all SCV phenotypes irrespective of their auxotrophism and genetic background revealed down-regulation of citric cycle activity as shown by the reduced 13C-incorporation into aspartate and glutamate with modified isotopolog profiles, as well as by down-regulation of acnA on the transcriptional level. A reduced metabolic status of all kinds of SCVs may explain the concordant major characteristics of the S. aureus SCV phenotypes regardless of the mechanism of their formation.
Conflict of Interest Statement
The Guest Associate Editor Thomas Dandekar declares that, despite having collaborated with authors Claudia Huber (Eisenreich group) and Wolfgang Eisenreich (Co-Topic Editor of this issue), the review process was handled objectively and no conflict of interest exists. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgment
We thank D. Kuhn, E. Leidig and M. Bach for excellent technical assistance. This work was supported by grants to Karsten Becker (BE 2546/1-2) and Wolfgang Eisenreich (EI 384/5-2) from the Deutsche Forschungsgemeinschaft (DFG) within the SPP1316.
References
Al Laham, N., Rohde, H., Sander, G., Fischer, A., Hussain, M., Heilmann, C., et al. (2007). Augmented expression of polysaccharide intercellular adhesin in a defined Staphylococcus epidermidis mutant with the small-colony-variant phenotype. J. Bacteriol. 189, 4494–4501. doi: 10.1128/JB.00160-07
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Balwit, J. M., van Langevelde, P., Vann, J. M., and Proctor, R. A. (1994). Gentamicin-resistant menadione and hemin auxotrophic Staphylococcus aureus persist within cultured endothelial cells. J. Infect. Dis. 170, 1033–1037. doi: 10.1093/infdis/170.4.1033
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Chatterjee, I., Kriegeskorte, A., Fischer, A., Deiwick, S., Theimann, N., Proctor, R. A., et al. (2008). In vivo mutations of thymidylate synthase (thyA) are responsible for thymidine-dependency in clinical small colony variants (TD-SCVs) of Staphylococcus aureus. J. Bacteriol. 190, 834–842. doi: 10.1128/JB.00912-07
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
David, M. Z., and Daum, R. S. (2010). Community-associated Methicillin-resistant Staphylococcus aureus: epidemiology and clinical consequences of an emerging epidemic. Clin. Microbiol. Rev. 23, 616–687. doi: 10.1128/CMR.00081-09
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Eylert, E., Schär, J., Mertins, S., Stoll, R., Bacher, A., Goebel, W., et al. (2008). Carbon metabolism of Listeria monocytogenes growing inside macrophages. Mol. Microbiol. 69, 1008–1017. doi: 10.1111/j.1365-2958.2008.06337.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Fraunholz, M., Bernhardt, J., Schuldes, J., Daniel, R., Hecker, M., and Sinha, B. (2013). Complete Genome Sequence of Staphylococcus aureus 6850, a Highly Cytotoxic and Clinically Virulent Methicillin-Sensitive Strain with Distant Relatedness to Prototype Strains. Genome Announc. 1:e00775-13. doi: 10.1128/genomeA.00775-13
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Gómez-González, C., Acosta, J., Villa, J., Barrado, L., Sanz, F., Orellana, M., et al. (2010). Clinical and molecular characteristics of infections with CO2-dependent small-colony variants of Staphylococcus aureus. J. Clin. Microbiol. 48, 2878–2884. doi: 10.1128/JCM.00520-10
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kahl, B. C., Belling, G., Reichelt, R., Herrmann, M., Proctor, R. A., and Peters, G. (2003). Thymidine-dependent small-colony variants of Staphylococcus aureus exhibit gross morphological and ultrastructural changes consistent with impaired cell separation. J. Clin. Microbiol. 41, 410–413. doi: 10.1128/JCM.41.1.410-413.2003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kohler, C., von Eiff, C., Liebeke, M., McNamara, P. J., Lalk, M., Proctor, R. A., et al. (2008). A defect in menadione biosynthesis induces global changes in gene expression in Staphylococcus aureus. J. Bacteriol. 190, 6351–6364. doi: 10.1128/JB.00505-08
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kohler, C., von Eiff, C., Peters, G., Proctor, R. A., Hecker, M., and Engelmann, S. (2003). Physiological characterization of a heme-deficient mutant of Staphylococcus aureus by a proteomic approach. J. Bacteriol. 185, 6928–6937. doi: 10.1128/JB.185.23.6928-6937.2003
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kriegeskorte, A., Konig, S., Sander, G., Pirkl, A., Mahabir, E., Proctor, R. A., et al. (2011). Small colony variants of Staphylococcus aureus reveal distinct protein profiles. Proteomics 11, 2476–2490. doi: 10.1002/pmic.201000796
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Kriegeskorte, A., Baum, C., Neumann, C., Seggewiss, J., Völker, W., Proctor, R. A., et al. (2012). “Physiology, morphology and fitness of Staphylococcus aureus thymidine-dependent small colony variants (TD-SCVs),” in 15th International Symposium on Staphylococci and Staphylococcal Infections (ISSSI) (Lyon).
Lannergård, J., von Eiff, C., Sander, G., Cordes, T., Seggewiss, J., Peters, G., et al. (2008). Identification of the genetic basis for clinical menadione-auxotrophic small-colony variant isolates of Staphylococcus aureus. Antimicrob. Agents Chemother. 52, 4017–4022. doi: 10.1128/AAC.00668-08
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Lowy, F. D. (1998). Staphylococcus aureus infections. N. Engl. J. Med. 339, 520–532. doi: 10.1056/NEJM199808203390806
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
O'Neill, A. J. (2010). Staphylococcus aureus SH1000 and 8325-4: comparative genome sequences of key laboratory strains in staphylococcal research. Lett. Appl. Microbiol. 51, 358–361. doi: 10.1111/j.1472-765X.2010.02885.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Proctor, R. A., von Eiff, C., Kahl, B. C., Becker, K., McNamara, P. J., Herrmann, M., et al. (2006). Small colony variants: a pathogenic form of bacteria that facilitates persistent and recurrent infections. Nat. Rev. Microbiol. 4, 295–305. doi: 10.1038/nrmicro1384
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Sachse, F., Becker, K., von Eiff, C., Metze, D., and Rudack, C. (2010). Staphylococcus aureus invades the epithelium in nasal polyposis and induces IL-6 in nasal epithelial cells in vitro. Allergy 65, 1430–1437. doi: 10.1111/j.1398-9995.2010.02381.x
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Seggewiss, J., Becker, K., Kotte, O., Eisenacher, M., Yazdi, M. R., Fischer, A., et al. (2006). Reporter metabolite analysis of transcriptional profiles of a Staphylococcus aureus strain with normal phenotype and its isogenic hemB mutant displaying the small-colony-variant phenotype. J. Bacteriol. 188, 7765–7777. doi: 10.1128/JB.00774-06
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Thomas, M. (1955). Studies on a CO2-dependent Staphylococcus aureus. J. Clin. Pathol. 8, 288–291. doi: 10.1136/jcp.8.4.288
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Tuchscherr, L., Heitmann, V., Hussain, M., Viemann, D., Roth, J., von Eiff, C., et al. (2010). Staphylococcus aureus small-colony variants are adapted phenotypes for intracellular persistence. J. Infect. Dis. 202, 1031–1040. doi: 10.1086/656047
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
von Eiff, C., Becker, K., Metze, D., Lubritz, G., Hockmann, J., Schwarz, T., et al. (2001). Intracellular persistence of Staphylococcus aureus small-colony variants within keratinocytes: a cause for antibiotic treatment failure in a patient with darier's disease. Clin. Infect. Dis. 32, 1643–1647. doi: 10.1086/320519
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
von Eiff, C., Heilmann, C., Proctor, R. A., Woltz, C., Peters, G., and Götz, F. (1997). A site-directed Staphylococcus aureus hemB mutant is a small-colony variant which persists intracellularly. J. Bacteriol. 179, 4706–4712.
von Eiff, C., Peters, G., and Becker, K. (2006). The small colony variant (SCV) concept - the role of staphylococcal SCVs in persistent infections. Injury 37, 26–33. doi: 10.1016/j.injury.2006.04.006
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text | Google Scholar
Keywords: S. aureus, SCV, metabolism, TCA
Citation: Kriegeskorte A, Grubmüller S, Huber C, Kahl BC, von Eiff C, Proctor RA, Peters G, Eisenreich W and Becker K (2014) Staphylococcus aureus small colony variants show common metabolic features in central metabolism irrespective of the underlying auxotrophism. Front. Cell. Infect. Microbiol. 4:141. doi: 10.3389/fcimb.2014.00141
Received: 14 July 2014; Accepted: 17 September 2014;
Published online: 21 October 2014.
Edited by:
Thomas Dandekar, University of Wuerzburg, GermanyReviewed by:
Matthew J. Wargo, University of Vermont College of Medicine, USAChristiane Wolz, University Tübingen, Germany
Agnes Fekete, University Wuerzburg, Germany
Copyright © 2014 Kriegeskorte, Grubmüller, Huber, Kahl, von Eiff, Proctor, Peters, Eisenreich and Becker. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Karsten Becker, Institute of Medical Microbiology, University of Münster, Domagkstraße 10, 48143 Münster, Germany e-mail: kbecker@uni-muenster.de
†Present address: Christof von Eiff, Pfizer Pharma GmbH, Berlin, Germany
 André Kriegeskorte
André Kriegeskorte Stephanie Grubmüller2
Stephanie Grubmüller2  Claudia Huber
Claudia Huber Barbara C. Kahl
Barbara C. Kahl Richard A. Proctor
Richard A. Proctor Wolfgang Eisenreich
Wolfgang Eisenreich Karsten Becker
Karsten Becker