- Bacterial Diseases Branch, Division of Vector Borne Diseases, National Center for Emerging and Zoonotic Infectious Diseases, Centers for Disease Control and Prevention, Fort Collins, CO, USA
Borrelia burgdorferi infection causes Lyme borreliosis in humans, a condition which can involve a systemic spread of the organism to colonize various tissues and organs. If the infection is left untreated by antimicrobials, it can lead to manifestations including, arthritis, carditis, and/or neurological problems. Identification and characterization of B. burgdorferi outer membrane proteins that facilitate cellular attachment and invasion to establish infection continue to be investigated. In this study, we sought to further define putative cell binding properties of surface-exposed B. burgdorferi proteins by observing whether cellular adherence could be blocked by antibodies. B. burgdorferi mixed separately with monoclonal antibodies (mAbs) against outer surface protein (Osp) A, OspC, decorin-binding protein (Dbp) A, BBA64, and RevA antigens were incubated with human umbilical vein endothelial cells (HUVEC) and human neuroglial cells (H4). B. burgdorferi treated with anti-OspA, -DbpA, and -BBA64 mAbs showed a significant decrease in cellular association compared to controls, whereas B. burgdorferi treated with anti-OspC and anti-RevA showed no reduction in cellular attachment. Additionally, temporal transcriptional analyses revealed upregulated expression of bba64, ospA, and dbpA during coincubation with cells. Together, the data provide evidence that OspA, DbpA, and BBA64 function in host cell adherence and infection mechanisms.
Introduction
Lyme borreliosis, or Lyme disease in humans, is the result of an infection with the spirochete Borrelia burgdorferi. This tick-borne illness typically exhibits a rash (termed erythema migrans) at the tick-bite site, and can progress to more severe conditions if left untreated. After introduction into the skin, the organism systemically disseminates and colonizes other tissues and organs ultimately resulting in arthritis, carditis, and/or neurological manifestations (Steere, 2001). The processes underlying the spirochete’s ability to spread and localize to host tissues leading to the pathology of Lyme disease are not fully defined.
Borrelia burgdorferi adapts to the varying environments encountered during its enzootic cycle through ticks and mammals by undergoing differential gene expression. For example, the organisms are relatively dormant in an unfed tick; however during tick feeding, B. burgdorferi becomes metabolically active by turning on genes encoding proteins essential for survival, replication, and transmission. Accordingly, once inside the mammalian host, B. burgdorferi regulates gene expression to facilitate infection. The number of genes expressed and gene products synthesized during mammalian infection is evidenced by the robust antibody response against a large number of borrelial antigens (Dressler et al., 1993). Some in vivo differentially expressed genes encode surface lipoproteins that have been identified as adhesins that mediate binding to molecules present in the extracellular matrix or on host cells of tissues (Coburn et al., 2005). Borrelial surface-exposed proteins decorin-binding protein (Dbp) A and B, BBK32, ErpX, RevA, Bgp, P66, BBB07, BmpA, CRASP-1, and outer surface protein (Osp) A, bind fibronectin, decorin, laminin, integrins, and other glycosaminoglycans and proteoglycans (Guo et al., 1998; Probert and Johnson, 1998; Pal et al., 2000; Parveen and Leong, 2000; Coburn and Cugini, 2003; Zambrano et al., 2004; Fischer et al., 2006; Rupprecht et al., 2006; Behera et al., 2008; Brissette et al., 2009a,b; Verma et al., 2009; Hallstrom et al., 2010).
Numerous studies have demonstrated borrelial adherence to and/or invasion of several cell types in vitro (Garcia-Monco et al., 1989; Szczepanski et al., 1990; Comstock and Thomas, 1991; Ma et al., 1991; Klempner et al., 1993; Kurtti et al., 1993; Girschick et al., 1996; Peters and Benach, 1997; Leong et al., 1998; Cinco et al., 2001; Fischer et al., 2003; Livengood and Gilmore, 2006; Wu et al., 2011). However, knowledge of specific processes mediated by known B. burgdorferi surface proteins for cell and tissue colonization, as well as the identification of novel proteins involved in host cell infections is lacking. In a previous study, we utilized global transcriptome analysis to examine B. burgdorferi gene expression during human host cell interactions as an initial step to identify proteins involved in cellular colonization, including adherence and invasion (Livengood et al., 2008). We hypothesized that genes upregulated in response to host cell signals may function in establishing B. burgdorferi infection. In this study, we examined whether B. burgdorferi binding to human cells could be blocked by antibodies directed against more well-characterized surface lipoproteins identified by the microarray (i.e., DbpA, BBA64, OspA, OspC, and RevA). Additionally, we measured transcription of these borrelial genes to observe the level of regulation in response to cellular interaction.
Materials and methods
Monoclonal Antibodies, Indirect Immunofluorescence (IFA) and Immunoblotting of Cultured B. burgdorferi
Monoclonal antibodies (mAbs) against BBA64, DbpA, and the anti-Bartonella protein were obtained from Barbara J. B. Johnson (CDC, Fort Collins, CO, USA). Anti-BBA64 and -DbpA were generated by recombinant protein immunization, and the anti-Bartonella mAb was generated by whole cell lysate immunization utilizing standard procedures for generating hybridomas in mice (Mbow et al., 2002). The anti-Bartonella mAb was reactive to a 70-kDa band on immunoblot against a rodent-derived Bartonella isolate. Anti-Rev and -OspC (B5) mAbs were generated by tick-bite inoculation of mice and have been described previously (Gilmore and Mbow, 1998; Mbow et al., 1999, 2002). Anti-OspA mAb H5332 was provided by Alan Barbour, UC-Irvine.
Immunofluorescent staining of cultured B. burgdorferi was performed as follows. B. burgdorferi low passage, infectious, clonal strain B31-A3 (Elias et al., 2002) was grown in complete Barbour-Stoenner-Kelly (BSK-II) medium at 34°C in capped tubes. Cultures were grown to mid-to-late logarithmic stage (approx. 5 × 107–1 × 108 organisms/ml), and 2 × 106 bacteria were spun onto Cytospin microscope slides using a Shandon Cytospin 4 (Thermo Electron Corporation, Waltham, MA, USA). After centrifugation, slides were air dried, incubated in blocking solution (2% bovine serum albumin in phosphate buffered saline pH 7.5 [BSA–PBS]) for 1 h at room temperature (rt), and then incubated with the specific mAb (1:75 dilution) for 1 h Slides were washed (3× with PBS) and stained with goat anti-mouse IgG Alexafluor 594 (Molecular Probes, Eugene, OR, USA) at a 1:75 dilution for 45 m. Slides were washed (3× with PBS) and probed with FITC-conjugated goat anti-Borrelia burgdorferi IgG (KPL, Gaithersburg, MD, USA) at a 1:75 dilution for 45 m. Slides were washed (3× with PBS), air dried, and overlaid with Prolong AntiFade (Molecular Probes) and coverslips prior to imaging by epifluorescent microscopy.
Immunoblotting was performed as follows. B. burgdorferi were collected from culture and washed in PBS prior to gel fractionation. Cell lysates were denatured by boiling for 10 min in Laemmli buffer containing 5% 2-mercaptoethanol and subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) on a 10–20% gradient gel followed by transfer to nitrocellulose filters according to standard procedures. Immunoblots were incubated with the appropriate mAb (1:5000 dilution) for 1 h, washed with PBS (3× at 5 min), followed by incubation with alkaline phosphatase conjugated goat anti-mouse IgG (1:2000) for 1 h. After PBS wash, the blot was developed colorimetrically with NBT (nitro-blue tetrazolium chloride) and BCIP (5-bromo-4-chloro-3′-indolyphosphate p-toluidine salt) substrate.
Mammalian Cell Culture
Human umbilical vein endothelial cells (HUVEC) and human neuroglial cells (H4) were obtained from American Type Culture Collection (Manassas, VA, USA; ATCC catalog numbers CRL-1730 and HTB-178 respectively). HUVECs were grown in F12-K medium supplemented with 0.1 mg/ml heparin, 0.03 mg/ml endothelial cell growth supplement, and 10% fetal bovine serum. The H4 cells were grown in Dulbecco’s modified eagle’s medium (DMEM) supplemented with 10% fetal bovine serum. Both cell lines were grown at 37°C in the presence of 5% CO2 in a humidified environment. Cells were harvested from confluent monolayers with 0.05% trypsin – 10 mM EDTA and enumerated on a hemocytometer cell counter.
Borrelia Burgdorferi-Cell Coincubation Association Assays
HUVEC and H4 cells were seeded at a concentration of 3 × 105 cells/chamber on Lab-Tek II CC chamber slide system (Nalge Nunc International, Rochester, NY, USA), and allowed to adhere overnight. Prior to addition to cells, individual B. burgdorferi aliquots, at a multiplicity of infection (MOI) of 40 (1.2 × 107), were preincubated (35°C shaking at 150 rpm for 30 m) with each anti-B. burgdorferi mAb or control in 0.5 ml volume. Following incubation with the appropriate antibody, B. burgdorferi were added to each cell well. The antibody concentration used was pre-determined empirically by a series of 10-fold dilutions for each mAb to ascertain the highest dilution of antibody that produced significant reduction in cell binding. Anti-BBA64, -DbpA, and -OspA were used at 1:500, 1:5000, and 1:5000 respectively. The coincubation experiments were performed in duplicate using these dilutions. Additionally, three control B. burgdorferi pre-cell incubation treatments were tested: (i) mAb suspension buffer only (50% glycerol/1× PBS); (ii) a non-Borrelia-specific mAb (anti-Bartonella antibody); and (iii) no antibody (with 1× PBS added to volume).
Following overnight coincubation of treated B. burgdorferi with human cells, slide chambers were gently washed 3× with PBS to remove non-adherent bacteria. Cells with associated bacteria were fixed on the slide chamber with 4% paraformaldehyde for 45 m at rt. Fixed cells were washed followed by blocking with 2% BSA-PBS for 1 h. Mammalian cells were stained with Alexa 594 Phalloidin (1:200 dilution; Molecular Probes) for 10 m at rt. Samples were washed, then cells were permeabilized with 2% BSA-PBS supplemented with 0.5% Triton X-100 for 45 m at rt. B. burgdorferi were stained with FITC-conjugated goat anti-B. burgdorferi (1:75 dilution) for 1 h.
Samples were visualized on a LSM 5 Pascal confocal microscope (Carl Zeiss, USA). Micrograph images were acquired at 100× and 250× magnification. B. burgdorferi associated with cells were enumerated microscopically. Counts were made for (1) total human cells/field, and (2) Borrelia associated with cells/field, with 30 individual fields observed and counted from each slide. From the sum of the 30 fields, the average number of B. burgdorferi associated per cell was determined. The percent reduction in cell attachment was calculated by the difference between the antibody-treated B. burgdorferi and the control treatment. Assays were performed in duplicate. Comparisons of the data were performed using a two-sample t-test. Differences were considered significant if p-value was ≤0.05.
RNA Isolation from B. burgdorferi-Associated Cells
HUVEC or H4 cells were grown to confluence in a 150 cm2 cell culture flask, trypsinized, and enumerated, from which 2 × 105 cells were seeded in six well cell culture plates and were allowed to adhere for 17 h at 37°C with 5% CO2. Log-phase B. burgdorferi cultures were centrifuged for 10 m at 9300 × g, with aliquots resuspended in F-12K or DMEM cell culture media and added to the HUVECs, H4 cells, or media alone at a MOI of 40 (7.8 × 107 bacteria). Plates were incubated at 34°C with 5% CO2. Cell wells were inoculated concurrently in triplicate and harvested for RNA isolation at 2-, 4-, 8-, 24-, and 48-h post-inoculation. At the appropriate time, cell culture media was removed, and the plate was washed 3× with 5 ml PBS to remove non-adherent bacteria. Cells with adherent bacteria were removed with cell scrapers in 300 μl RNAlater (Ambion, Austin, TX, USA). Media from the cell-free Borrelia wells were centrifuged at 9300 × g for 5 min, washed 2× with 5 ml PBS, and resuspended in 300 μl RNAlater. RNAlater samples were stored overnight at 4°C and then at −20°C until the RNA was purified. Prior to RNA extraction, the samples were centrifuged at 16,000 × g for 10 m, the RNAlater was decanted, followed by total RNA extraction using RNAqueous RNA isolation kit (Ambion). Contaminating DNA was removed using the TURBO DNA-free kit (Ambion). RNA samples were tested for contaminating DNA by PCR prior to cDNA synthesis. Total RNA was quantified by spectrophotometry and RNA integrity was analyzed using an Agilent 2100 Bioanalyzer with Agilent RNA Pico Reagents (Agilent Technologies, Boulder CO).
Quantitative Reverse Transcriptase PCR (qRT-PCR)
Purified total RNA (100 ng) isolated from the B. burgdorferi-associated cells was used to synthesize cDNA using SuperScript II Reverse Transcriptase (Invitrogen, Carlsbad, CA, USA) and random decamers. Real-time PCR was performed with TaqMan Universal PCR Master Mix (Applied Biosystems) using flaB, bba64, dbpA, and ospA TaqMan probe and primers described previously (Livengood et al., 2008). Real-time PCR reactions, performed in triplicate per sample, contained 1 μl cDNA, 1 μM 5′ primer, 1 μM 3′ primer, 0.1 μM probe, and 1× TaqMan Universal PCR Master mixture in a total volume of 12.5 μl. Amplification parameters consisted of 1 cycle at 95°C for 10 m and 50 cycles of 95°C for 30 s and 60°C for 1 m, with data collection after each cycle in a BioRad iCycler (BioRad, Hercules, CA, USA). The iCycler software determined crossing threshold (CT) values. B. burgdorferi gene expression during coincubation with cells was determined relative to borrelial gene expression in cell-free tissue culture media using the 2−ΔΔCT method (Livak and Schmittgen, 2001), and was normalized against the constitutively expressed flaB gene.
Results
Protein Synthesis by in vitro Cultivated B. burgdorferi
Cultured B. burgdorferi was assessed for the production of OspA, DbpA, BBA64, OspC, and RevA prior to antibody treatment and coincubation with tissue culture cells. Analysis of cultured organisms by IFA demonstrated in vitro production of the individual proteins (Figure 1A). Immunoblot analysis of cultured borrelial lysates also showed protein production (Figure 1B).
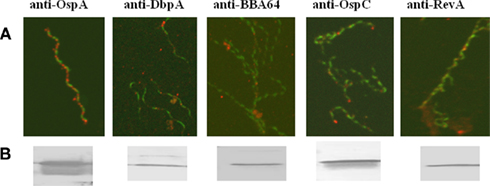
Figure 1. Production of proteins from cultured B. burgdorferi. (A) Representative images of immunofluorescent dual staining of organisms with the specific mAb (noted above panels) and anti-B. burgdorferi antibody. Punctate staining of surface proteins are labeled red; whole cell B. burgdorferi are stained green. (B) Immunoblots of B. burgdorferi lysates using the specified mAbs.
Coincubation of Antibody-Treated B. burgdorferi with HUVEC and H4 Cells
Borrelia burgdorferi were preincubated with the mAbs for 30 m prior to placement on HUVEC and H4 cells. Following the antibody incubation, a sample of the spirochetes were visualized under darkfield microscopy to determine viability. B. burgdorferi treated with each mAb were motile indicating that the antibody treatment was not borreliacidal.
An experiment was performed to find the least concentrated mAb solution that produced significant differences (p < 0.05) in cell binding from the no antibody control treatment. We considered that higher dilutions of mAb would provide less chance for steric hindrance that could lead to non-specific blocking. The optimal dilutions were determined to be 5 × 10−3 for the anti-OspA and -DbpA mAbs, and 5 × 10−2 for the anti-BBA64 antibody. Anti-RevA and anti-OspC mAbs, at all dilutions tested, demonstrated no significant inhibition of binding to HUVEC or H4 cells when compared to the no antibody control (data not shown).
Additionally, there were no significant differences between numbers of B. burgdorferi attached to host cells following each of the control treatments, i.e., Borrelia preincubated with antibody buffer only, Borrelia preincubated with non-specific antibody, and Borrelia preincubated with only PBS (data not shown). Therefore, the control data reported (as “without” or “no antibody”) in the text reflect counts obtained with the non-specific anti-Bartonella antibody pretreatment control.
Table 1 shows associated B. burgdorferi/100 cells with and without specific mAb treatment and the calculated reduction in B. burgdorferi cellular attachment. B. burgdorferi treated with anti-OspA, -DbpA, and -BBA64 antibodies demonstrated significant decreases in HUVEC and H4 cell binding when compared to Borrelia treated with the control. (As mentioned above, treatment with anti-OspC and anti-RevA had no effect on binding). The reduction in attachment between antibody treatments in separate experiments ranged from 30.6 to 55.2% for both HUVEC and H4 cells. The average B. burgdorferi/100 cells counted from 30 fields in each experiment were significantly different between antibody-treated Borrelia and controls (p < 0.001). An additive effect in attachment reduction when all three antibodies were combined was not observed. Figure 2 shows images of representative fields of B. burgdorferi binding to HUVECs and H4 cells respectively, with and without antibody treatment, allowing a visual inspection of the diminished attachment.
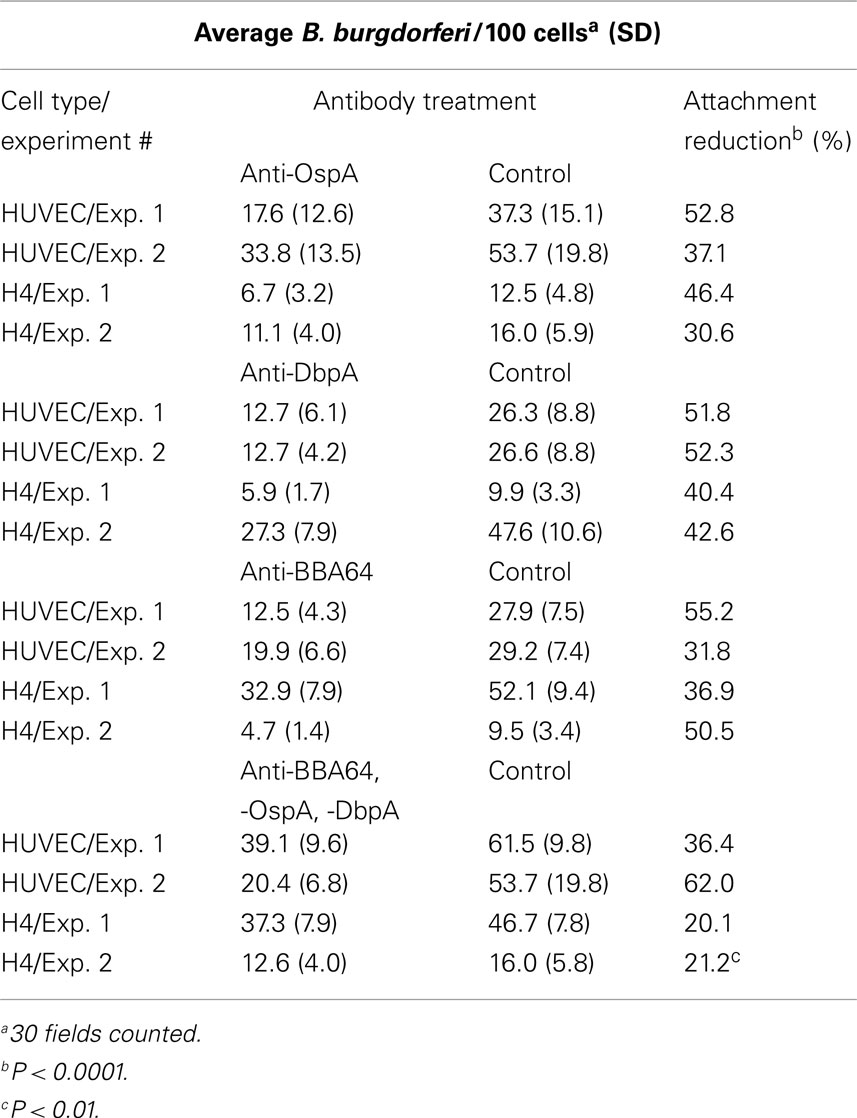
Table 1. Reduction in B. burgdorferi binding to HUVEC and H4 cells following treatment with anti-B. burgdorferi surface protein antibodies.
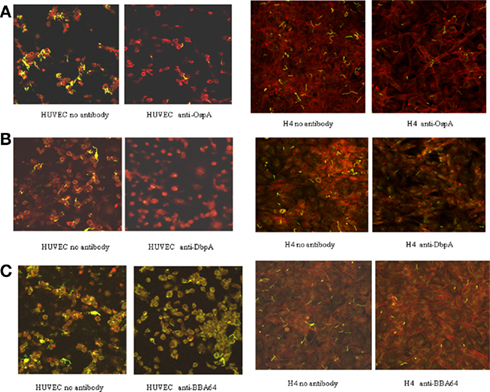
Figure 2. Confocal microscopy of fluorescent-stained B. burgdorferi bound to HUVEC and H4 cells. Panels on the left show Borrelia attachment with no antibody treatment (i.e., the anti-Bartonella control); panels on the right show Borrelia attachment following incubation with anti-surface protein mAb. (A) anti-OspA; (B) anti-DbpA; (C) anti-BBA64.
OspA, DbpA, and BBA64 Gene Expression during Coincubation with Host Cells
Antibodies directed against OspA, DbpA, and BBA64 attenuated the ability of B. burgdorferi to bind to host cells in vitro suggesting that these surface proteins may play a role in establishing infection. Therefore, we hypothesized that the genes encoding these proteins may be upregulated during cellular interaction. We employed qRT-PCR to monitor B. burgdorferi gene expression temporally throughout the incubation period with the tissue culture cells. We found that in both H4 and HUVEC interactions, dbpA, bba64, and ospA expression were generally elevated compared to B. burgdorferi incubation in cell-free environment.
dbpA expression was the highest of the three genes in both HUVEC and H4 incubations. At 2-h post-inoculation, gene expression was approximately 150-fold higher in HUVEC than in cell-free media (Figure 3A). dbpA expression continued to be elevated at approximately 20-fold at 4-, 8-, 24-, and 48-h post-inoculation in HUVECs compared to the control. dbpA also was upregulated in H4 cells throughout the incubation period, with a peak around 60-fold at 48 h (Figure 3A).
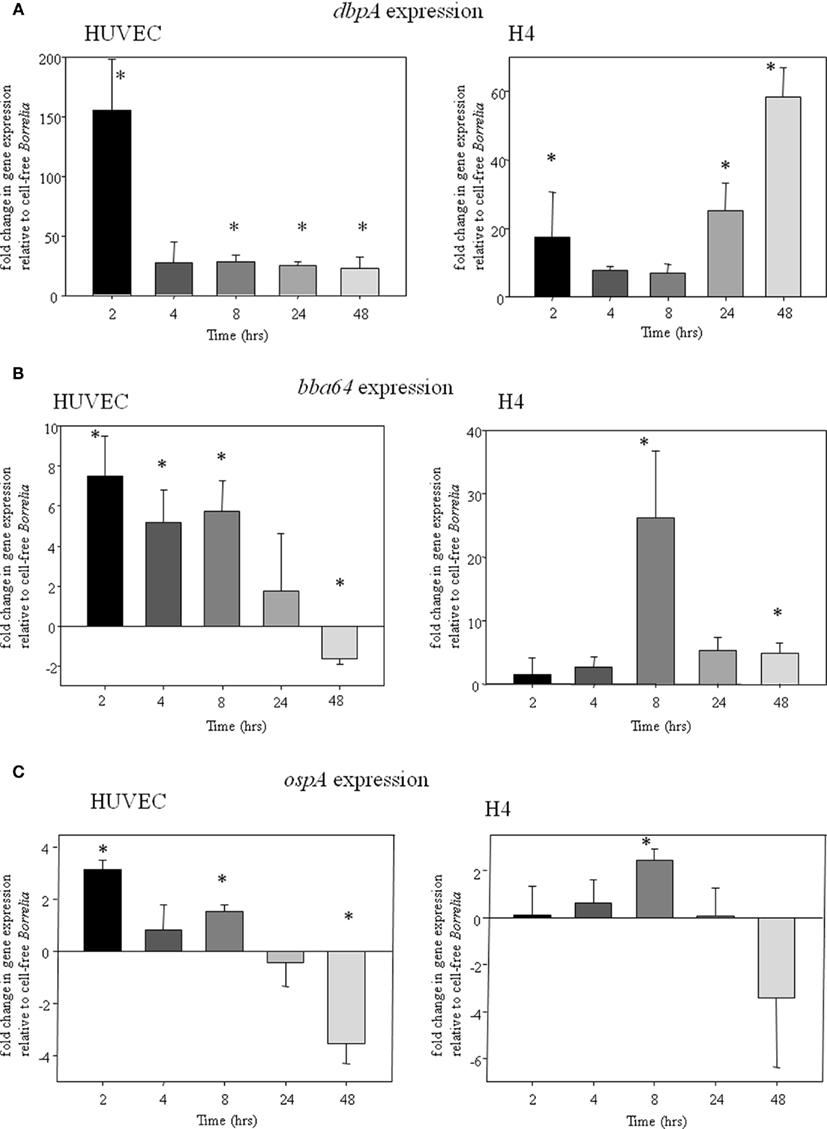
Figure 3. (A) dbpA gene expression in HUVEC (left graph) and H4 cells (right graph); (B) bba64 gene expression in HUVEC (left graph) and H4 cells (right graph); (C) ospA gene expression in HUVEC (left graph) and H4 (right graph).
bba64 expression measured at 2- to 8-h post-inoculation with HUVECs was between approximately five- to sevenfold higher than in cell-free media (Figure 3B). At 24-h post-inoculation, expression was detected but was not significantly increased compared to B. burgdorferi in cell-free media. However, by 48 h, expression had decreased below the cell-free level. In H4 cells, bba64 expression was detected early, but not at significantly higher levels than in cell-free organisms. However, expression increased several-fold by 8-h post-inoculation, and remained elevated compared with the cell-free control at 48-h post-inoculation (Figure 3B).
ospA expression was detectable and slightly elevated in HUVECs in early time points post-inoculation when compared to expression in cell-free media. At 2 h into the incubation period, an approximate threefold increase in expression was observed. At 4-h post-inoculation, the expression level had decreased from 2 h, although a significant increase from cell-free expression was detected at 8 h. Later in the incubation, at 24 and 48 h, ospA expression had decreased to levels below that in cell-free media. A similar pattern was observed in H4 cells where there was no significant difference between expression in cell-free conditions, except at 8 h incubation whereby there was an approximate twofold increase (Figure 3C).
Discussion
Colonization of host tissues by B. burgdorferi is a key factor in the pathogenesis of Lyme borreliosis. However, the mechanisms by which B. burgdorferi interacts with host cells to facilitate attachment, invasion, and colonization are poorly understood. To gain insight into B. burgdorferi genes that encode proteins required for human cell interactions, we hypothesized in an earlier study that genes upregulated during host cell infection may have roles in adherence, invasion, and colonization. In that study, we performed a global analysis of B. burgdorferi gene expression by microarray following coincubation of Borrelia with human neuroglial cells and found several genes to be upregulated during the cellular interaction (Livengood et al., 2008). Among the upregulated genes identified by the microarray study were ospA, dbpA, and bba64. Other studies have shown ospC and revA to encode products that functioned in establishing mammalian infection and in fibronectin-binding respectively (Grimm et al., 2004; Brissette et al., 2009a). Therefore, we initiated the current study to determine if mAbs directed against these surface proteins could prevent or reduce the establishment of host cell attachment, and if so, assay expression of the genes during cellular association.
We first determined that cultured B. burgdorferi produced the target proteins prior to antibody treatment and cell incubation. Second, after mixing B. burgdorferi and the mAbs we ensured that the antibody was not bactericidal and that the spirochetes were still motile. With these requirements satisfied, B. burgdorferi incubated with and without specific antibody were coincubated in tissue culture with the human host cells. Three of the five anti-surface protein antibodies, anti-OspA, anti-DbpA, and anti-BBA64, significantly reduced B. burgdorferi binding to H4 and HUVECs (Table 1; Figure 2).
In an earlier study by Comstock et al. (1993), a mAb directed against OspA (9B3D) was shown to inhibit binding to HUVECs, therefore our anti-OspA treatment served as a positive control and allowed us to corroborate the finding with a mAb directed against OspA. The antibodies directed against RevA or OspC had no inhibitory effect on B. burgdorferi binding, suggesting these proteins do not play a role in attachment to HUVEC or H4 cells. RevA is expressed during mammalian infection and has recently been shown to be a fibronectin-binding protein, but its function is unknown (Gilmore and Mbow, 1998; Brissette et al., 2009a). OspC has been studied more extensively and is required by B. burgdorferi to establish infection in the mammalian host (Grimm et al., 2004). Because mAbs specific for a single epitope on the protein were used, we cannot discount that polyclonal antibodies directed against other determinants of OspC or RevA could provide a blocking effect. However, the anti-OspC mAb used here has been shown to protect against B. burgdorferi challenge when passively administered (Mbow et al., 1999). Therefore the failure of this mAb to inhibit cell adherence suggests that OspC does not function in cellular interactions (at least for these cell types) to promote infection. The lack of effect on cell binding with these two antibodies also provided evidence that the reduction in attachment measured with the anti-OspA, -DbpA, and -BBA64 antibodies was not due to non-specific inhibition.
The results of the antibody blocking experiments suggested that OspA, BBA64, and DbpA can mediate B. burgdorferi attachment to human cells, and that their production may be necessary for cell and tissue colonization. Therefore, we measured B. burgdorferi gene expression during coincubation with the host cells, and found that dbpA, ospA, and bba64 expression were elevated during interactions with both cell types.
dbpA was highly upregulated, throughout the 48-h experiment, when B. burgdorferi were incubated with HUVECs and H4 cells. DbpA is an adhesin that binds decorin. a protein linked to the glycosaminoglycan (GAG) dermatan sulfate/chondroitin sulfate and is most commonly associated with collagen, but is present in many tissues (Guo et al., 1995, 1998). Proteoglycans are receptors for B. burgdorferi adherence to several cell types, with specific classes of GAGs playing key roles in the spirochetal binding activity (Leong et al., 1998). Fischer et al. (2003) showed that transformation of a non-adherent strain of B. burgdorferi lacking the dbpBA operon with dbpA could restore binding to human epithelial cells. Our observation of dbpA upregulation in response to interactions with host cells, combined with the reduction in cell binding by antibody reinforces findings from earlier studies linking DbpA production with cell attachment, tissue colonization and host immune resistance (Liang et al., 2004; Blevins et al., 2008; Shi et al., 2008b; Weening et al., 2008).
bba64 was also expressed during B. burgdorferi interactions with HUVEC and H4 cells. Recently we demonstrated that bba64 was necessary for mammalian infection via tick transmission, and that bba64 is upregulated in ticks during bloodmeal ingestion (Gilmore et al., 2010; Patton et al., 2011). In addition, we have shown bba64 expression in mouse tissues throughout persistent infection, suggesting a role in host pathogenesis, possibly by functioning in the establishment and maintenance of the organism in various tissue (Gilmore et al., 2007, 2008). The upregulation of bba64 expression when coincubated with human cells implicates BBA64 as a multifunctional protein involved in tick transmission and mammalian host cell interactions.
Previous studies have implicated OspA in cell adherence even though ospA is downregulated in the tick during feeding, and is not expressed in the host in the acute phases of infection. Rupprecht et al. (2006) demonstrated that OspA from B. garinii, a B. burgdorferi sensu lato strain most frequently associated with Lyme neuroborreliosis in Europe, mediates adherence to neuronal cells in vitro. Also, Comstock et al. (1993) showed that a mAb against OspA could inhibit borrelial association with HUVECs. In our study, ospA was expressed, and significantly upregulated early in response to HUVEC interaction, and at 8-h post-inoculation in H4 cells. The early expression of ospA may reflect an unknown function relating to host cell attachment, with downregulation of expression later after the spirochetes have established cellular colonization. However, because ospA is minimally or not expressed at the time of mammalian entry, the role of OspA in mediating cellular interactions remains to be determined.
Generation of B. burgdorferi mutant isolates, whereby ospA, dbpA, and bba64 have been inactivated, have led to studies to define the role of these genes in host infectivity and pathogenesis. Attenuated tissue colonization and host persistence has been shown in mice injected with mutant B. burgdorferi lacking dbpA, and decorin-deficient mouse strains demonstrated resistance to experimental infection by both needle and tick bite (Brown et al., 2001; Blevins et al., 2008; Shi et al., 2008a; Weening et al., 2008). In contrast, ospA- and bba64-knockout mutants (when introduced by needle inoculation) were not impaired in their ability to infect or persist in mice (Yang et al., 2004; Battisti et al., 2008; Maruskova et al., 2008; Gilmore et al., 2010). Despite being dispensible for mouse infection via needle inoculation of cultured organisms, bba64 is highly expressed by wild-type organisms in several tissues during persistent infection in vivo (Gilmore et al., 2008). It is becoming clear that B. burgdorferi binding and colonization of host cell tissues is a complex process mediated by numerous adherence components produced by B. burgdorferi, which may contribute to functional redundancy during infection. Perhaps such redundancy provides a survival advantage to host-adapted B. burgdorferi to evade host immunity, or a combination of proteins are regulated to work synergistically to optimally colonize the host.
In conclusion, we have demonstrated that antibodies directed against OspA, DbpA, and BBA64 can significantly reduce the binding ability of B. burgdorferi to human endothelial and neuroglial cells. Additionally, we have shown that ospA, bba64, and dbpA are expressed and upregulated during times of B. burgdorferi-host cell interactions in vitro. Collectively, these findings infer that these outer surface proteins contribute to borrelial attachment and host cell colonization. Future studies will address the mechanisms employed by these gene products in cellular infection by utilizing mutant strains with inactivated dbpA, bba64, ospA to interface with human cells in tissue culture. Additionally, we seek to identify novel B. burgdorferi genes that are upregulated during interactions with human cells as a means to understand and characterize the response of this pathogen to host components during infection.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors acknowledge Jill Livengood for her valuable comments and input during the acquisition of data for this manuscript. We thank Steve Sviat, Barbara J. B. Johnson, and Alan Barbour for the mAbs used in this study.
References
Battisti, J. M., Bono, J. L., Rosa, P. A., Schrumpf, M. E., Schwan, T. G., and Policastro, P. F. (2008). Outer surface protein A protects Lyme disease spirochetes from acquired host immunity in the tick vector. Infect. Immun. 76, 5228–5237.
Behera, A. K., Durand, E., Cugini, C., Antonara, S., Bourassa, L., Hildebrand, E., Hu, L. T., and Coburn, J. (2008). Borrelia burgdorferi BBB07 interaction with integrin alpha3beta1 stimulates production of pro-inflammatory mediators in primary human chondrocytes. Cell. Microbiol. 10, 320–331.
Blevins, J. S., Hagman, K. E., and Norgard, M. V. (2008). Assessment of decorin-binding protein A to the infectivity of Borrelia burgdorferi in the murine models of needle and tick infection. BMC Microbiol. 8, 82.
Brissette, C. A., Bykowski, T., Cooley, A. E., Bowman, A., and Stevenson, B. (2009a). Borrelia burgdorferi RevA antigen binds host fibronectin. Infect. Immun. 77, 2802–2812.
Brissette, C. A., Verma, A., Bowman, A., Cooley, A. E., and Stevenson, B. (2009b). The Borrelia burgdorferi outer-surface protein ErpX binds mammalian laminin. Microbiology 155, 863–872.
Brown, E. L., Wooten, R. M., Johnson, B. J., Iozzo, R. V., Smith, A., Dolan, M. C., Guo, B. P., Weis, J. J., and Hook, M. (2001). Resistance to Lyme disease in decorin-deficient mice. J. Clin. Invest. 107, 845–852.
Cinco, M., Cini, B., Murgia, R., Presani, G., Prodan, M., and Perticarari, S. (2001). Evidence of involvement of the mannose receptor in adhesion of Borrelia burgdorferi to monocyte/macrophages. Infect. Immun. 69, 2743–2747.
Coburn, J., and Cugini, C. (2003). Targeted mutation of the outer membrane protein P66 disrupts attachment of the Lyme disease agent, Borrelia burgdorferi, to integrin alphavbeta3. Proc. Natl. Acad. Sci. U.S.A. 100, 7301–7306.
Coburn, J., Fischer, J. R., and Leong, J. M. (2005). Solving a sticky problem: new genetic approaches to host cell adhesion by the Lyme disease spirochete. Mol. Microbiol. 57, 1182–1195.
Comstock, L. E., Fikrig, E., Shoberg, R. J., Flavell, R. A., and Thomas, D. D. (1993). A monoclonal antibody to OspA inhibits association of Borrelia burgdorferi with human endothelial cells. Infect. Immun. 61, 423–431.
Comstock, L. E., and Thomas, D. D. (1991). Characterization of Borrelia burgdorferi invasion of cultured endothelial cells. Microb. Pathog. 10, 137–148.
Dressler, F., Whalen, J. A., Reinhardt, B. N., and Steere, A. C. (1993). Western blotting in the serodiagnosis of Lyme disease. J. Infect. Dis. 167, 392–400.
Elias, A. F., Stewart, P. E., Grimm, D., Caimano, M. J., Eggers, C. H., Tilly, K., Bono, J. L., Akins, D. R., Radolf, J. D., Schwan, T. G., and Rosa, P. (2002). Clonal polymorphism of Borrelia burgdorferi strain B31 MI: implications for mutagenesis in an infectious strain background. Infect. Immun. 70, 2139–2150.
Fischer, J. R., LeBlanc, K. T., and Leong, J. M. (2006). Fibronectin binding protein BBK32 of the Lyme disease spirochete promotes bacterial attachment to glycosaminoglycans. Infect. Immun. 74, 435–441.
Fischer, J. R., Parveen, N., Magoun, L., and Leong, J. M. (2003). Decorin-binding proteins A and B confer distinct mammalian cell type-specific attachment by Borrelia burgdorferi, the Lyme disease spirochete. Proc. Natl. Acad. Sci. U.S.A. 100, 7307–7312.
Garcia-Monco, J. C., Fernandez-Villar, B., and Benach, J. L. (1989). Adherence of the Lyme disease spirochete to glial cells and cells of glial origin. J. Infect. Dis. 160, 497–506.
Gilmore, R. D. Jr., Howison, R. R., Dietrich, G., Patton, T. G., Clifton, D. R., and Carroll, J. A. (2010). The bba64 gene of Borrelia burgdorferi, the Lyme disease agent, is critical for mammalian infection via tick bite transmission. Proc. Natl. Acad. Sci. U.S.A. 107, 7515–7520.
Gilmore, R. D. Jr., Howison, R. R., Schmit, V. L., and Carroll, J. A. (2008). Borrelia burgdorferi expression of the bba64, bba65, bba66, and bba73 genes in tissues during persistent infection in mice. Microb. Pathog. 45, 355–360.
Gilmore, R. D. Jr., Howison, R. R., Schmit, V. L., Nowalk, A. J., Clifton, D. R., Nolder, C., Hughes, J. L., and Carroll, J. A. (2007). Temporal expression analysis of the Borrelia burgdorferi paralogous gene family 54 genes BBA64, BBA65, and BBA66 during persistent infection in mice. Infect. Immun. 75, 2753–2764.
Gilmore, R. D. Jr., and Mbow, M. L. (1998). A monoclonal antibody generated by antigen inoculation via tick bite is reactive to the Borrelia burgdorferi Rev protein, a member of the 2.9 gene family locus. Infect. Immun. 66, 980–986.
Girschick, H. J., Huppertz, H. I., Russmann, H., Krenn, V., and Karch, H. (1996). Intracellular persistence of Borrelia burgdorferi in human synovial cells. Rheumatol. Int. 16, 125–132.
Grimm, D., Tilly, K., Byram, R., Stewart, P. E., Krum, J. G., Bueschel, D. M., Schwan, T. G., Policastro, P. F., Elias, A. F., and Rosa, P. A. (2004). Outer-surface protein C of the Lyme disease spirochete: a protein induced in ticks for infection of mammals. Proc. Natl. Acad. Sci. U.S.A. 101, 3142–3147.
Guo, B. P., Brown, E. L., Dorward, D. W., Rosenberg, L. C., and Hook, M. (1998). Decorin-binding adhesins from Borrelia burgdorferi. Mol. Microbiol. 30, 711–723.
Guo, B. P., Norris, S. J., Rosenberg, L. C., and Hook, M. (1995). Adherence of Borrelia burgdorferi to the proteoglycan decorin. Infect. Immun. 63, 3467–3472.
Hallstrom, T., Haupt, K., Kraiczy, P., Hortschansky, P., Wallich, R., Skerka, C., and Zipfel, P. F. (2010). Complement regulator-acquiring surface protein 1 of Borrelia burgdorferi binds to human bone morphogenic protein 2, several extracellular matrix proteins, and plasminogen. J. Infect. Dis. 202, 490–498.
Klempner, M. S., Noring, R., and Rogers, R. A. (1993). Invasion of human skin fibroblasts by the Lyme disease spirochete, Borrelia burgdorferi. J. Infect. Dis. 167, 1074–1081.
Kurtti, T. J., Munderloh, U. G., Krueger, D. E., Johnson, R. C., and Schwan, T. G. (1993). Adhesion to and invasion of cultured tick (Acarina: Ixodidae) cells by Borrelia burgdorferi (Spirochaetales: Spirochaetaceae) and maintenance of infectivity. J. Med. Entomol. 30, 586–596.
Leong, J. M., Wang, H., Magoun, L., Field, J. A., Morrissey, P. E., Robbins, D., Tatro, J. B., Coburn, J., and Parveen, N. (1998). Different classes of proteoglycans contribute to the attachment of Borrelia burgdorferi to cultured endothelial and brain cells. Infect. Immun. 66, 994–999.
Liang, F. T., Brown, E. L., Wang, T., Iozzo, R. V., and Fikrig, E. (2004). Protective niche for Borrelia burgdorferi to evade humoral immunity. Am. J. Pathol. 165, 977–985.
Livak, K. J., and Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the  method. Methods 25, 402–408.
method. Methods 25, 402–408.
Livengood, J. A., and Gilmore, R. D. Jr. (2006). Invasion of human neuronal and glial cells by an infectious strain of Borrelia burgdorferi. Microbes Infect. 8, 2832–2840.
Livengood, J. A., Schmit, V. L., and Gilmore, R. D. Jr. (2008). Global transcriptome analysis of Borrelia burgdorferi during association with human neuroglial cells. Infect. Immun. 76, 298–307.
Ma, Y., Sturrock, A., and Weis, J. J. (1991). Intracellular localization of Borrelia burgdorferi within human endothelial cells. Infect. Immun. 59, 671–678.
Maruskova, M., Esteve-Gassent, M. D., Sexton, V. L., and Seshu, J. (2008). Role of the BBA64 locus of Borrelia burgdorferi in early stages of infectivity in a murine model of Lyme disease. Infect. Immun. 76, 391–402.
Mbow, M. L., Gilmore, R. D. Jr., Stevenson, B., Golde, W. T., Piesman, J., and Johnson, B. J. (2002). Borrelia burgdorferi-specific monoclonal antibodies derived from mice primed with Lyme disease spirochete-infected Ixodes scapularis ticks. Hybrid. Hybridomics 21, 179–182.
Mbow, M. L., Gilmore, R. D. Jr., and Titus, R. G. (1999). An OspC-specific monoclonal antibody passively protects mice from tick-transmitted infection by Borrelia burgdorferi B31. Infect. Immun. 67, 5470–5472.
Pal, U., de Silva, A. M., Montgomery, R. R., Fish, D., Anguita, J., Anderson, J. F., Lobet, Y., and Fikrig, E. (2000). Attachment of Borrelia burgdorferi within Ixodes scapularis mediated by outer surface protein A. J. Clin. Invest. 106, 561–569.
Parveen, N., and Leong, J. M. (2000). Identification of a candidate glycosaminoglycan-binding adhesin of the Lyme disease spirochete Borrelia burgdorferi. Mol. Microbiol. 35, 1220–1234.
Patton, T. G., Dietrich, G., Dolan, M. C., Piesman, J., Carroll, J. A., and Gilmore, R. D. Jr. (2011). Functional analysis of the Borrelia burgdorferi bba64 gene product in murine infection via tick infestation. PLoS ONE 6, e19536.
Peters, D. J., and Benach, J. L. (1997). Borrelia burgdorferi adherence and injury to undifferentiated and differentiated neural cells in vitro. J. Infect. Dis. 176, 470–477.
Probert, W. S., and Johnson, B. J. (1998). Identification of a 47 kDa fibronectin-binding protein expressed by Borrelia burgdorferi isolate B31. Mol. Microbiol. 30, 1003–1015.
Rupprecht, T. A., Koedel, U., Heimerl, C., Fingerle, V., Paul, R., Wilske, B., and Pfister, H. W. (2006). Adhesion of Borrelia garinii to neuronal cells is mediated by the interaction of OspA with proteoglycans. J. Neuroimmunol. 175, 5–11.
Shi, Y., Xu, Q., McShan, K., and Liang, F. T. (2008a). Both decorin-binding proteins A and B are critical for the overall virulence of Borrelia burgdorferi. Infect. Immun. 76, 1239–1246.
Shi, Y., Xu, Q., Seemanaplli, S. V., McShan, K., and Liang, F. T. (2008b). Common and unique contributions of decorin-binding proteins A and B to the overall virulence of Borrelia burgdorferi. PLoS ONE 3, e3340.
Szczepanski, A., Furie, M. B., Benach, J. L., Lane, B. P., and Fleit, H. B. (1990). Interaction between Borrelia burgdorferi and endothelium in vitro. J. Clin. Invest. 85, 1637–1647.
Verma, A., Brissette, C. A., Bowman, A., and Stevenson, B. (2009). Borrelia burgdorferi BmpA is a laminin-binding protein. Infect. Immun. 77, 4940–4946.
Weening, E. H., Parveen, N., Trzeciakowski, J. P., Leong, J. M., Hook, M., and Skare, J. T. (2008). Borrelia burgdorferi lacking DbpBA exhibits an early survival defect during experimental infection. Infect. Immun. 76, 5694–5705.
Wu, J., Weening, E. H., Faske, J. B., Hook, M., and Skare, J. T. (2011). Invasion of eukaryotic cells by Borrelia burgdorferi requires beta1 integrins and src kinase activity. Infect. Immun. 79, 1338–1348.
Yang, X. F., Pal, U., Alani, S. M., Fikrig, E., and Norgard, M. V. (2004). Essential role for OspA/B in the life cycle of the Lyme disease spirochete. J. Exp. Med. 199, 641–648.
Keywords: Borrelia burgdorferi, cell attachment, gene expression
Citation: Schmit VL, Patton TG and Gilmore RD Jr. (2011) Analysis of Borrelia burgdorferi surface proteins as determinants in establishing host cell interactions. Front. Microbio. 2:141. doi: 10.3389/fmicb.2011.00141
Received: 19 May 2011;
Accepted: 14 June 2011;
Published online: 01 July 2011.
Edited by:
Robert Heinzen, NIH/NIAID-RML, USAReviewed by:
Michael F. Minnick, The University of Montana, USAEdward Shaw, Oklahoma State University, USA
Copyright: © 2011 Schmit, Patton and Gilmore. This is an open-access article subject to a non-exclusive license between the authors and Frontiers Media SA, which permits use, distribution and reproduction in other forums, provided the original authors and source are credited and other Frontiers conditions are complied with.
*Correspondence: Robert D. Gilmore Jr., Division of Vector Borne Diseases, Centers for Disease Control and Prevention, 3150 Rampart Rd., Fort Collins, CO 80521, USA. e-mail: rbg9@cdc.gov
