
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Hum. Neurosci. , 22 November 2013
Sec. Cognitive Neuroscience
Volume 7 - 2013 | https://doi.org/10.3389/fnhum.2013.00779
This article is part of the Research Topic Near-infrared spectroscopy (NIRS) in functional research of prefrontal cortex View all 20 articles
Listening to music engages the whole brain, thus stimulating cognitive performance in a range of non-purely musical activities such as language and memory tasks. This article addresses an ongoing debate on the link between music and memory for words. While evidence on healthy and clinical populations suggests that music listening can improve verbal memory in a variety of situations, it is still unclear what specific memory process is affected and how. This study was designed to explore the hypothesis that music specifically benefits the encoding part of verbal memory tasks, by providing a richer context for encoding and therefore less demand on the dorsolateral prefrontal cortex (DLPFC). Twenty-two healthy young adults were subjected to functional near-infrared spectroscopy (fNIRS) imaging of their bilateral DLPFC while encoding words in the presence of either a music or a silent background. Behavioral data confirmed the facilitating effect of music background during encoding on subsequent item recognition. fNIRS results revealed significantly greater activation of the left hemisphere during encoding (in line with the HERA model of memory lateralization) and a sustained, bilateral decrease of activity in the DLPFC in the music condition compared to silence. These findings suggest that music modulates the role played by the DLPFC during verbal encoding, and open perspectives for applications to clinical populations with prefrontal impairments, such as elderly adults or Alzheimer’s patients.
Listening to music engages the whole brain through a diverse set of perceptive and cognitive operations, and equally diverse neural substrates (Altenmüller, 2003). As most of these neural substrates also intervene in other activities, it is increasingly believed that music can benefit non-musical abilities, and most notably language (Abbott and Avins, 2006; Patel, 2011). In particular, there is an ongoing debate in the field of music and cognitive stimulation on whether music can be used to enhance verbal memory. On the one hand, music is a complex auditory stimulus which evolves through time and which has a strong emotional impact (Blood and Zatorre, 2001; Salimpoor et al., 2013). As such, music can provide considerable additional cues which are likely to enrich the encoding of an event. On the other hand, musical information was also claimed to negatively affect memory by attracting participants’ attention away from the information to be remembered, generating a dual task situation with poorer memory performance than in a silent situation (Racette and Peretz, 2007; Moussard et al., 2012). In the last 20 years, several studies were conducted in order to understand when and how music can have a positive effect on memory. Research on western music indicates that musical training (Chan et al., 1998; Ho et al., 2003; Franklin et al., 2008) and also simple exposure to music leads to benefits on short- and long-term verbal memory in healthy and clinical populations (Balch et al., 1992; Balch and Lewis, 1996). In 1994, Wallace showed that text is better recalled when heard as a song rather than speech, suggesting that musical context can assist in learning and retrieving words. In clinical settings, short (i.e., music played as a background in a memory task) and long-lasting (i.e., in a music-therapy program) auditory stimulations with music were both shown to improve category fluency in a verbal fluency task in both healthy elderly and Alzheimer’s patients (Thompson et al., 2005), speech content and fluency in patients with dementia (Brotons and Koger, 2000), and verbal memory in stroke patients (Särkämö et al., 2008). Additionally, verbal material is more efficiently retrieved when sung than spoken in multiple sclerosis (Thaut et al., 2005), aphasics (Racette et al., 2006), and Alzheimer’s patients (Simmons-Stern et al., 2010).
Such evidence suggests that music provides contextual cues that contribute to episodic memory processes. Episodic memory (Tulving, 1972) enables conscious recollection of personal experiences and events from the past (Wheeler et al., 1997). Encoding is a crucial aspect of episodic memory and it is tightly related to the contribution of contextual factors such as location, time, prevailing conditions, and converging multisensory and emotional stimuli (Eich, 1985; Hamann, 2001; Kensinger and Corkin, 2003; Hupbach et al., 2008). Neuroimaging and behavioral data have clearly shown that the capacity to retrieve correct information depends on its successful encoding (e.g., Prince et al., 2005; Hertzog et al., 2010). Furthermore, richer contexts enhance the encoding of contextual information associated to an item and can be subsequently used as mnemonic cues during retrieval, facilitating the access to the target item (Lövdén et al., 2002; Kroneisen and Erdfelder, 2011). It has been shown that enriching the context of encoding through, e.g., enacted encoding (Lövdén et al., 2002) or with emotional valence stimuli (see Hamann, 2001 for a review) can enhance memory performance at retrieval. It is therefore possible that the greatest value of music for memory is to provide mnemonic processes with a particularly rich and helpful context during the encoding phase of episodic memory.
Memory encoding and retrieval processes are supported by a broad brain network that involves the medio-temporal and posterior parietal areas, the hippocampus, and the prefrontal cortex (PFC), the latter being particularly important for episodic memory (Spaniol et al., 2009; Manenti et al., 2012). Different sub-regions of the PFC are recruited by different mnemonic processes: according to the hemispheric encoding/retrieval asymmetry (HERA) model (Tulving et al., 1994), left PFC activation is greater for encoding than retrieval, while right PFC activation is greater for retrieval than encoding. Although the PFC sub-region most constantly associated with memory in neuroimaging studies is the ventrolateral prefrontal cortex (VLPFC – BA 44-45-47), the dorsolateral-prefrontal cortex (DLPFC – BA 9 and 46) has recently gained importance for the specific investigation of memory encoding processes. In particular, it has been shown that the DLPFC, mainly in the left hemisphere, plays a crucial role for organizational, associative (Murray and Ranganath, 2007; Ranganath, 2010) and semantic (Innocenti et al., 2010) memory encoding. As discussed by Blumenfeld and Ranganath (2007), DLPFC activation seems to be more specifically sensitive to demands for organizational processing and it may support long term memory by building associations among items that are active in memory.
Functional near-infrared spectroscopy (fNIRS) is an optical neuroimaging technique that can non-invasively monitor cortical tissue oxygenation (oxygenated-O2Hb and deoxygenated-HHb hemoglobin concentration changes) during cognitive, motor, and sensory stimulation (Jobsis, 1977; Ferrari and Quaresima, 2012). In the last 20 years, the use of fNIRS in cognitive neuroscience has constantly increased (Ferrari and Quaresima, 2012). In the field of memory research in particular, fNIRS studies have revealed an increase in PFC oxygenation patterns (i.e., an increase in O2Hb and concomitant decrease in HHb concentrations) during working memory and attention tasks in healthy and clinical populations (see Cutini et al., 2012 for a review). However, the current literature only has a limited number of fNIRS studies (Kubota et al., 2006; Matsui et al., 2007; Okamoto et al., 2011) investigating episodic encoding-retrieval processes. In the field of music cognition, a few fNIRS studies were recently conducted in order to investigate the emotional response to music (Moghimi et al., 2012a; Moghimi S et al., 2012b). However, no fNIRS study has yet looked at a possible role of music in memory encoding.
The previous fNIRS studies that have documented facilitating factors on memory encoding, e.g., strategies to memorize words (Matsui et al., 2007), or pharmacological stimulants such as methylphenidate (Ramasubbu et al., 2012), have repeatedly shown that such factors deactivate, rather than more greatly activate, regions of the PFC – as if they were “less-demanding” (Matsuda and Hiraki, 2004, 2006). Similar reductions of PFC activation were shown using fMRI when there were strong semantic associations between words (Addis and McAndrews, 2006). These evidence suggest that facilitatory cues (e.g., strategies, pharmacological stimulant, strong semantic associations) during verbal encoding could result in less involvement of high cognitive functions mediated by PFC regions (such as DLPFC) known to be usually crucial during memory encoding processes.
The present study addresses the music and memory debate using a source memory paradigm: participants were asked to memorize both lists of words and the context/source (either music or silence) in which words were encoded. The critical new point of the present study was to assess whether the presence of background music during the encoding of verbal material results in different memory-specific cortical patterns of activations than episodic encoding in silence. We used fNIRS to monitor the DLPFC bilaterally during the encoding of verbal material with or without background music context. Our hypothesis is that music may enhance verbal encoding by providing a helpful context which can facilitate organizational, associative, and semantic processes. If so, such an effect of music on memory encoding processes should be linked to both behavioral performance and PFC activity. More specifically, we consider a facilitatory effect of music during verbal encoding should result in a better recognition performance and deactivation of DLPFC activity during the music encoding condition compared to the silent condition.
Twenty-two young healthy students at University of Burgundy (11 female, mean age 23.5 ± 4.3 years) took part in the experiment in exchange of course credits. All the participants were right-handed, non-musicians, French-native speakers, and reported having normal or corrected-to-normal vision. None were taking medication known to affect the central nervous system. Informed written consent was obtained from all participants prior to taking part in the experiment. The study was anonymous and fully obeyed to the Helsinki Declaration, Convention of the Council of Europe on Human Rights and Biomedicine.
Subjects were seated in a chair in front of a computer in a quiet, dim room. Each participant was subjected to a memory encoding task while their PFC activation was monitored using fNIRS neuroimaging and then behaviorally tested in a retrieval task. After the eight fNIRS probe-set was adjusted on the forehead scalp overlaying the DLPFC (see fNIRS section below for a description) and the in-ear headphones inserted, subjects were informed that they would be presented with different lists of words with two different auditory contexts: music or silence. They were asked to memorize both the lists of words and the context in which words were encoded.
Verbal stimuli consisted of 42 taxonomically unrelated concrete nouns selected from the French “Lexique” database (New et al., 2004, http://www.lexique.org). They were randomly divided into 6 lists of words (7 words each list, 21 words for each encoding condition), equated for word length and occurrence frequency. In the music encoding condition, the background music used in all blocks was an upbeat, acoustic jazz piece (“If you see my mother” by Sidney Bechet), chosen for its positive valence and medium arousal quality.
The encoding phase consisted of three blocks of “music encoding” and three blocks of “silence encoding” intermixed with 30-s rest periods. In each block, seven words were displayed successively in the presence of a music or silence auditory context. The audio stimulation started 15 s before the first word was displayed, continued during the sequential display of words, and ended 15 s after the last word. Words displayed in each block were paced at 4 s per word, amounting to 28 s for the sequential presentation of seven words. Each block therefore had a duration of 58 s (15 s context, 28 s words, 15 s context), and was followed by a 30 s rest (silent) between each block (Figure 1). The order of music/silence blocks was counterbalanced, as well as the order of word lists and the order of words in the lists. During the rest periods subjects were instructed to try to relax and not to think about the task any longer; in contrast, during the context-only phases of the blocks (i.e., silence and music blocks), participants were instructed to concentrate on a fixation cross on the screen and to prepare/focus on the task. The entire encoding phase, together with fNIRS recording, took about 10 min.
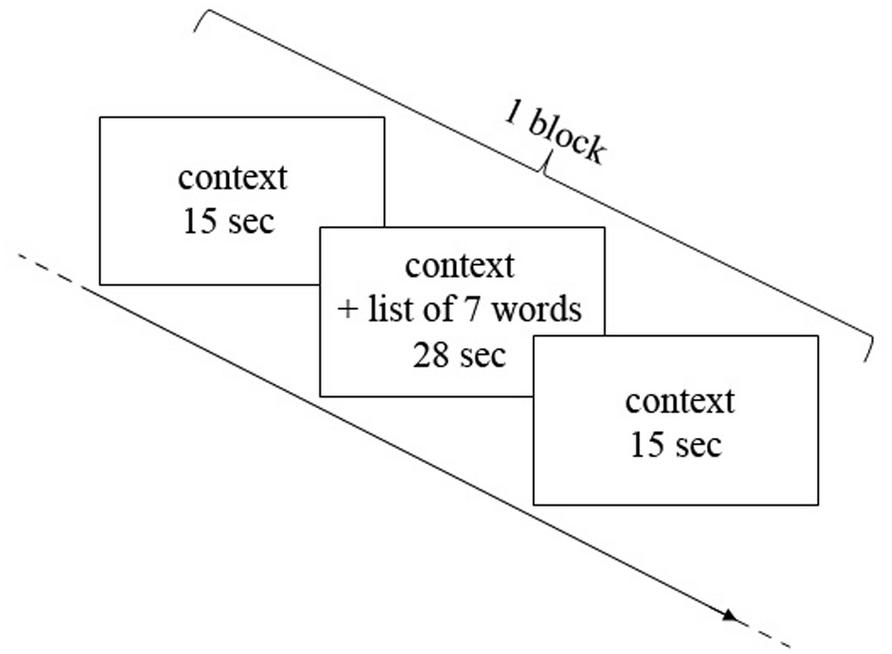
FIGURE 1. Representation of one block of encoding. Each block consisted in 15 s of context alone (music or silence in the earphones), then 28 s of context and words encoding (seven words for each block, 4 s for each word) and then again 15 s of context alone.
Prior to the retrieval phase, subjects performed two 5-min interference tasks: a “X-O” letter-comparison task (Salthouse et al., 1997) and a “plus–minus” task (Jersild, 1927; Spector and Biederman, 1976). Subjects were then tested for item and source memory recognition. We used item-memory and source-memory tasks (Glisky et al., 1995) in order to evaluate the subjects’ memory for the context of encoding. The retrieval test included the 42 words presented previously, together with 42 new words which were lure items matched for word length and occurrence frequency. For each word, subjects were asked to judge if they had already seen the word before (yes/no button on the keyboard; item-memory task). If they believed they had, they were asked to indicate in which context they saw the word (music/silence/I don’t know; source-memory task). The presentation of task instructions and stimuli as well as the recording of behavioral responses were controlled by the E-Prime software (Psychology Software Tools, Inc.) running on a laptop with a 15′′ monitor.
An eight-channel fNIRS system (Oxymon MkIII, Artinis Medical Systems B.V., The Netherlands) was used to measure the concentration changes of O2Hb and HHb (expressed in micromoles) using an age-dependent constant differential path-length factor given by 4.99 + 0.0067*(age 0.814; Duncan et al., 1996). Data was acquired at a sampling frequency of 10 Hz. The eight fNIRS optodes (four emitters and four detectors) were placed symmetrically over the dorsal part of the PFC (Brodmann Areas 46 and 9, EEG electrodes AF7/8, F5/6, F3/4, and AF3/4 of the international 10/10 system; Okamoto et al., 2004; Jurcak et al., 2007), and the distance between each emitter and detector was fixed to 3.5 cm (Figure 2).
To optimize signal-to-noise ratio during the fNIRS recording, the eight optodes were masked from ambient light by a black plastic cap that was kept in contact with the scalp with elastic straps, and all cables were suspended from the ceiling to minimize movement artifacts (Cui et al., 2011). During data collection, O2Hb and HHb concentration changes were displayed in real time, and the signal quality and the absence of movement artifacts were verified.
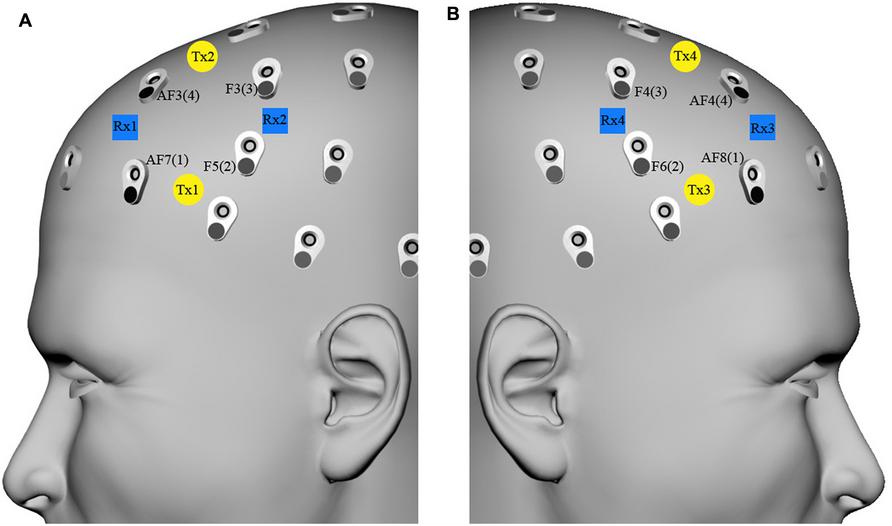
FIGURE 2. fNIRS optode localization on the forehead scalp region overlying the dorsal part of the prefrontal cortex. fNIRS transmitters [(A) Tx1–Tx2, (B) Tx3–Tx4, yellow circles) and receivers [(A) Rx1–Rx2, (B) Rx3–Rx4, blue squares) were placed on the (A) and (B) forehead scalp region, which corresponded to AF7/8, F5/6, F3/4, and AF3/4 EEG channels (international 10/10 system), respectively renamed left/right channels 1, 2, 3, and 4.
Each subject’s item- and source-memory accuracy (hit) rates (number of hits for each condition during the yes/no recognition) as well as false alarms were calculated for both the silence and music conditions. To examine source memory, we analyzed the proportion of correct source judgments among item-memory hits. A paired t-test was used to compare the item- and source- memory scores between the silence and music conditions. One sample t-tests were used to ascertain that all the scores were significantly above chance.
For each of the eight fNIRS measurement points, the O2Hb and HHb signals were first low-pass filtered to eliminate task-irrelevant systemic physiological oscillations (fifth order digital Butterworth filter with cutoff frequency 0.1 Hz).
In order to ascertain the DLPFC activation during the word encoding task as compared to the rest phase, we first ran a complete timecourse analysis on the O2Hb and HHb signals using a 2(music/silence condition) × 2(left/right hemisphere) × 4(optodes) × 13(successive measures of concentrations, averaged over 5 s windows with the last 10 s of the rest phase as baseline) repeated-measures ANOVA, on which Fisher’s LSD post hoc comparisons determine which steps of the O2Hb and HHb time course showed significant increase/decrease of O2Hb and HHb as compared to the baseline point set during the rest phase (Figure 3).
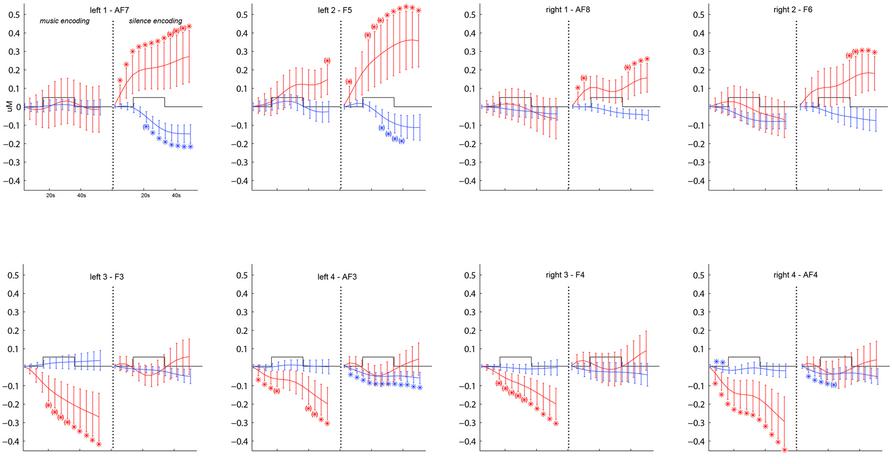
FIGURE 3. Grand-average (±SEM) time course of prefrontal cortex O2Hb (red lines) and HHb (blue lines) concentration changes (vertical axis) over the left and right hemisphere during memory encoding (horizontal axis: time) for the silence (right side) and music (left side) conditions. * and (*) show, respectively, significant (p < 0.05) and marginally significant (0.05 < p < 0.09) time points compared to baseline obtained by post hoc Fisher’s LSD comparisons.
In order to determine the amount of activation during the encoding phase for the two conditions, data in each of the six experimental blocks was baseline corrected using the mean of the O2Hb and HHb signals during the first 5 s of each block (i.e., during context-only phase, before the start of word encoding). We then sample-to-sample averaged (i.e., 10 samples/s) the baseline-corrected signals over the three blocks of each condition, yielding one average music and silence O2Hb and HHb signal per participant. We then computed the maximum O2Hb (max- O2Hb) value and the minimum HHb (min-HHb) value over the 28 s stimulus window (i.e., from t = 15 s to t = 42 s), for both the music and silence average block of each participant. The peak concentrations (max-O2Hb, min-HHb) were analyzed using a repeated measure MANOVA with 2(music/silence condition) × 2(left/right hemisphere) repeated factors and optodes (4) as a multivariate. The significance level was set at p < 0.05.
Both the item and source memory scores were significantly above chance (one sample t-test, p < 0.003), demonstrating that participants did not encounter strong difficulties to remember the specific context in which words were presented.
There was a statistically significant difference in item recognition performance between the music (mean= 18.36, SD= 2.84) and silence [mean= 16.59, SD= 3.98; t(21) = 2.63, p = 0.016] conditions, with improved recognition in the musical condition. However, there was no significant difference (p > 0.05) in source memory performance between music (0.67 ± 0.22) and silence (0.68 ± 0.22) conditions.
Figure 3 shows the grand average time course of PFC O2Hb and HHb concentration changes at each of the eight fNIRS channels in the music and silence encoding conditions. The repeated-measure ANOVA on the O2Hb and HHb timecourse series revealed a main effect of condition (F = 8.130, p = 0.01), corresponding to significantly greater O2Hb increases bilaterally in the silence than music condition. Although the increases in O2Hb are visible bilaterally during the silence condition (especially in bilateral channels 1 and 2 as shown by post hoc LSD Fisher comparisons), together with a decrease in HHb (in particular for left channels 1, 2 and right channel 2), the music condition was associated with a strong bilateral decrease of O2Hb (underlined by significant LSD Fisher post hoc comparisons especially for bilateral channels 3 and 4).
Figure 4 shows the group mean of max-O2Hb and min-HHb values recorded at each of the eight fNIRS channels on the PFC for the silence and music conditions. The repeated-measure MANOVA on max-O2Hb values revealed a statistically significant main effect of condition [F(4,18) = 4.207, p = 0.008], with greater O2Hb increases bilaterally in the silence than music condition, and a significant main effect of channel laterality [F(4,18) = 5.006, p = 0.003], with greater O2Hb increases in the left hemisphere regardless of condition. Although there was no effect of condition for min-HHb values, which is typical with several other NIRS studies (e.g., Matsui et al., 2007; Okamoto et al., 2011), there was a significant effect of laterality [F(4,18) = 3.783, p = 0.013], with greater values in the right hemisphere (which is coherent with smaller O2Hb values, i.e., overall smaller O2Hb increases in the right hemisphere).
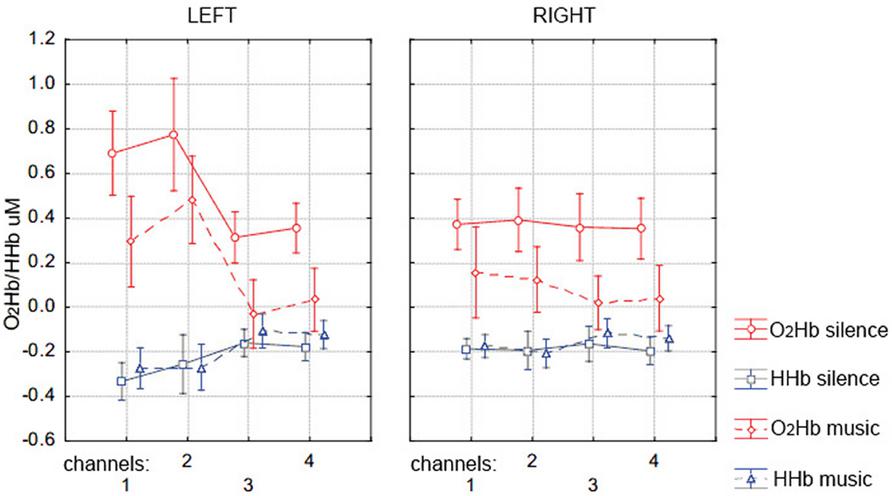
FIGURE 4. Mean (±SD) of the prefrontal cortex max-O2Hb (red lines) and min-HHb (blue lines) concentration values over the left (channels 1, 2, 3, and 4, corresponding to EEG channels AF7, F5, AF3, F3) and right (channels 1, 2, 3, and 4, corresponding to EEG channels AF8, F6, F4, AF4) hemisphere during memory encoding for the silence (solid lines) and music (dotted lines) condition.
The present study shows that a background musical context during the encoding of verbal material modulates the activation of the DLPFC and, at the same time, facilitates the retrieval of the encoded material. Despite a few recent studies (e.g., Racette and Peretz, 2007; Moussard et al., 2012) reporting a perturbing effect of music on the memorization of verbal material, a consistent part of the literature (e.g., Balch et al., 1992; Wallace, 1994; Balch and Lewis, 1996; Chan et al., 1998; Brotons and Koger, 2000; Ho et al., 2003; Thaut et al., 2005; Thompson et al., 2005; Racette et al., 2006; Franklin et al., 2008; Särkämö et al., 2008; Simmons-Stern et al., 2010) claims that music can have a positive effect on memory in both healthy and clinical populations. However, most of these studies had remained on a behavioral level. The critical new point of the present study was to also track the brain activation response during the encoding phase with fNIRS: we found that improved word recognition coincides with reduced DLPFC activation in musical encoding compared to silence encoding.
Our behavioral results showed an effect of music on subsequent item recognition memory performance, although this did not extend to a source memory performance. We found that music played during encoding facilitates item recognition. The role of background music in learning and memory tasks is still an open and debated question in the literature (e.g., Schellenberg, 2003, 2005; De Groot, 2006; Peterson and Thaut, 2007; Jäncke and Sandmann, 2010). Research in music cognition is increasingly aware of the fact that it is necessary, rather than stating a general and reliable positive effect of music, to disentangle which experimental paradigm can lead to memory improvements through music, when and with whom. Our results are in line with previous studies on healthy subjects (Balch et al., 1992; Wallace, 1994; Balch and Lewis, 1996; De Groot, 2006) and clinical populations (Brotons and Koger, 2000; Ho et al., 2003; Thaut et al., 2005; Thompson et al., 2005; Racette et al., 2006; Franklin et al., 2008; Särkämö et al., 2008; Simmons-Stern et al., 2010) which showed a positive role of music in verbal memory encoding. Consistent with the view that context is crucial during episodic memory encoding, our findings support the idea that music provides rich and helpful contextual cues that are useful for subsequent item recognition.
The novel finding of the study was that DLPFC activation was significantly higher during the silence than music encoding condition (Figures 3 and 4). As predicted in our hypothesis, the facilitatory effect of music during verbal encoding resulted not only in better recognition performance, but also deactivation of DLPFC activity. On the one hand, encoding PFC activity in the silent condition followed the classical hemodynamic response to neuronal activation, showing a bilateral increase of O2Hb together with a decrease of HHb as compared to baseline. This result confirms the involvement of the DLPFC in episodic memory encoding (Blumenfeld and Ranganath, 2007; Murray and Ranganath, 2007; Innocenti et al., 2010). On the other hand, encoding in the music condition showed a bilateral reversed PFC hemodynamic response (with a sustained decrease in O2Hb and minimal change in HHb), which only returned to baseline at the end of each music block (see Figure 3). This result suggests that the DLPFC was deactivated during word encoding in the musical context and that music can strongly modulate the activity of the bilateral DLPFC. Similar PFC deactivation have already been shown by fNIRS studies investigating human cognition (Matsuda and Hiraki, 2006, 2004), and specifically in verbal learning tasks when subjects were helped to memorize words by a given strategy (Matsui et al., 2007) or by a pharmacological stimulant (Ramasubbu et al., 2012).
However, to the best of our knowledge, none of the previous fNIRS studies which investigated memory processes or music perception reported music-specific PFC deactivation. In the present study, this PFC deactivation during memory encoding with a musical context could be the manifestation of our hypothesis that music plays a facilitating, less-demanding role for the PFC during word encoding.
fNIRS analysis of the maximum O2Hb concentration values reached during word encoding in the music and silence conditions revealed also a significant main effect of lateralization. As predicted, we found greater PFC activation (represented by O2Hb increases) in the left than the right hemisphere during the entire encoding phase (especially for channels 1 and 2, see Figure 4). This result is in line with the hemispheric left prefrontal asymmetry during the encoding of verbal material, as predicted by the HERA model (Tulving et al., 1994; Nyberg et al., 1996), and confirms the feasibility of fNIRS neuroimaging for the study of long-term memory processes (Kubota et al., 2006; Matsui et al., 2007; Okamoto et al., 2011).
It is important to discuss the possible mechanisms by which music may act on the PFC during memory encoding tasks. The PFC, specifically the DLPFC, is known to be recruited during tasks demanding organizational (Blumenfeld and Ranganath, 2007) and relational inter-item processing during encoding (Murray and Ranganath, 2007). Therefore, one possible interpretation of the deactivation of the PFC (i.e., O2Hb decrease) during music in the present study is that music helps to generate inter-item and item-source relationships, without demanding high-cognitive PFC processes. Investigating the correlation between PFC activity and semantic associations during word encoding with fMRI, Addis and McAndrews (2006) found that greater semantic associations correlated with reduced activity in the inferior frontal gyrus (IFG) region of the PFC. A recent fNIRS study by Ramasubbu et al. (2012) also supports this explanation. The authors gave methylphenidate (a central nervous system stimulant) or placebo to subjects and measured PFC activation during a working memory task (N-back). They found a reduction in PFC O2Hb from baseline together with better behavioral performance, which the authors suggested was due to methylphenidate improving neuronal efficiency or signal–noise ratio during the memory task. In the present study, the decreased PFC activity observed during the music condition could therefore indicate better neuronal efficiency.
The musical context may afford efficient mnemonic strategies to bind items between one another, and/or to bind items to music, so that less PFC activity is required to drive these associations. In line with the idea of music as an help for cognitive functions which could lead to a deactivation of PFC activity, it has been recently shown how exposure to consonant music improve performance during a Stroop task, suggesting that music may help overcoming cognitive interference (Masataka and Perlovsky, 2013). So how could music represent a facilitatory factor particularly for words encoding? Previous EEG studies underlined how few seconds of music can influence the semantic and conceptual processes of words, showing that both music and language can prime the meaning of a word and determine physiological indices of semantic processes (Koelsch et al., 2004; Daltrozzo and Schön, 2009a,b). It is therefore possible that this semantic priming could also be reflected in easier associations and bindings between items when background music is present. Further investigations on organizational strategies during verbal encoding with music may confirm this explanation and shed new lights on music-verbal memory cognitive processes.
Another possible explanation of the music-specific PFC deactivation is an increase of attentional mechanisms in the music condition. Music is known to modulate attentional processes (Janata et al., 2002; Janata and Grafton, 2003; see also Masataka and Perlovsky, 2013) and previous fNIRS studies reported that the more attention the subjects put to a task, the more greatly rCBF (Mazoyer et al., 2002; Geday and Gjedde, 2009) and O2Hb concentrations (Matsuda and Hiraki, 2004, 2006) were decreased in the PFC. This second interpretation would be in line with previous behavioral studies which attributed improved cognitive performance in the presence of a musical background to higher amounts of arousal and attention (Foster and Valentine, 2001; Thompson et al., 2005; Patel, 2011). However, in apparent conflict with this interpretation, a considerable amount of literature claims the importance of the PFC in attentional processes, mainly for the maintenance and mental manipulation of memory contents (see Ptak, 2012 for a review). In the present study, we observed that PFC O2Hb increase seems to precede the word encoding phase by a few seconds in the most lateral fNIRS channels (i.e., left channels 1 and 2, corresponding to EEG channels AF7 and F3, Figure 3), and this, even in the silence condition (Figure 3). This may indicate that attentional processes are already in full use when encoding in silence, and put a limit on the potential of even further recruitment of attention specific to music.
Finally, we should also note that it has been repeatedly observed that music-related processing typically recruits more widely distributed networks of cortical and subcortical areas than non-musical verbal function (Halpern, 2001; Parsons, 2001; Peretz, 2002; Altenmüller, 2003; Patel, 2011). If so, PFC deactivation in the music condition could also reflect broader network recruitment during word encoding with music. Further research is needed to test this hypothesis.
fNIRS data interpretation of the present study must be done bearing in mind certain limitations. First, recent studies suggest that caution should be exercised when applying fNIRS to infer PFC activation: the task-evoked changes occurring in forehead skin perfusion could represent an overestimation of the cortical changes as measured by fNIRS. Recent reports have raised a question against the assumption that PFC O2Hb/HHb changes originated only from the cortical hemodynamic response (Kohno et al., 2007; Gagnon et al., 2011, 2012; Takahashi et al., 2011; Kirilina et al., 2012). Furthermore, as previously described, fNIRS acquisitions in the present study were limited to eight channels covering the bilateral DLPFC. So, it is not possible to know whether other cortical areas were involved during episodic encoding, especially for the music condition. Despite these limitations, several studies have shown fNIRS feasibility for the study of cognitive processes (Cutini et al., 2012), and this study for the first time applied fNIRS to investigate if and how music can help memory during episodic encoding.
An important perspective for further research is to apply fNIRS monitoring during the retrieval phase. Indeed, research on episodic memory during the past century has demonstrated that a complete understanding of how memories are formed requires appreciation of the many cognitive and neurobiological processes that constitute encoding and retrieval, as well as the interaction among these two stages (Brown and Craik, 2000). The behavioral and fNIRS data we obtained lead us to wonder about what is also happening during retrieval. Further studies with multichannel fNIRS systems during both encoding and retrieval phases are needed to determine which regions are more activated and to clarify how music could act on long term memory processes.
Another interesting perspective for further studies is to extend our paradigm to applications in older adults or patients with dementia. Several studies have highlighted that memory impairments in normal aging as well as several types of dementia (e.g., Alzheimer’s disease) are often linked to impairments or damage in frontal lobe functions (e.g., see Maillet and Rajah, 2012 for a review). Our results suggest that music helps verbal encoding by facilitating associative and organizational processes (i.e., generate inter-item and item-source relationships) without demanding the high-cognitive PFC processes which are usually required. At the same time, fNIRS is a non-invasive technique and its features allow it to be used also with special populations by preserving good ecological settings (Cutini et al., 2012; Ferreri et al., in press). Further fNIRS investigations on normal and pathological aging could therefore be pivotal for better understanding of how music can be used as a tool in rehabilitation of memory disorders.
In conclusion, we have shown that background music context during the encoding of verbal material modulates the activation of the PFC during encoding and, at the same time, facilitates the retrieval of the encoded material. This opens interesting perspectives on how music could act on the PFC of subjects with memory disorders for whom the prefrontal lobe is hypo-activated, impaired or damaged, such as older adults or Alzheimer’s patients.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
This work was supported by the European Project EBRAMUS (European BRAin and MUSic) ITN – Grant Agreement Number 218357 and the Conseil Régional de Bourgogne. Authors also thank Chris Moulin for insightful comments on the manuscript.
Abbott, E., and Avins, K. (2006). “Music, health and well-being,” in Complementary and Alternative Medicine for Older Adults, eds E. R. MacKenzie and B. Rakel (New York: Springer Publishing Company), 97–100.
Addis, D. R., and McAndrews, M. P. (2006). Prefrontal and hippocampal contributions to the generation and binding of semantic associations during successful encoding. Neuroimage 33, 1194–1206. doi: 10.1016/j.neuroimage.2006.07.039
Altenmüller, E. O. (2003). “How many music centers are in the brain?” in The Cognitive Neuroscience of Music, eds I. Peretz and R. J. Zatorre (Oxford, NY: Oxford University Press), 346–356. doi: 10.1093/acprof:oso/9780198525202.003.0022
Balch, W. R., and Lewis, B. S. (1996). Music-dependent memory: the roles of tempo change and mood mediation. J. Exp. Psychol. Learn. Mem. Cogn. 22, 1354–1363. doi: 10.1037/0278-7393.22.6.1354
Balch, W. R., Bowman, K., and Mohler, L. A. (1992). Music-dependent memory in immediate and delayed word recall. Mem. Cognit. 20 21–28. doi: 10.3758/BF03208250
Blood, A. J., and Zatorre, R. J. (2001). Intensely pleasurable responses to music correlate with activity in brain regions implicated in reward and emotion. Proc. Natl. Acad. Sci. U.S.A. 98, 11818–11823. doi: 10.1073/pnas.191355898
Blumenfeld, R. C., and Ranganath, C. (2007). Prefrontal cortex and long-term memory encoding: an integrative review of findings from neuropsychology and neuroimaging. Neuroscientist 13, 280–291. doi: 10.1177/1073858407299290
Brotons, M., and Koger, S. M. (2000). The impact of music therapy on language functioning in dementia. J. Music Ther. 37 183–195.
Brown, S. C., and Craik, F. I. M. (2000). “Encoding and retrieval of information,” in The Oxford Handbook of Memory, eds E. Tulving and F. I. M. Craik (New York: Oxford University Press), 93–107.
Chan, A. S., Ho, Y., and Cheung, M. (1998). Music training improves verbal memory. Nature 396 128. doi: 10.1038/24075
Cui, X., Bray, S., Bryant, D. M., Glover, G. H., and Reiss, A. L. (2011). A quantitative comparison of NIRS and fMRI across multiple cognitive tasks. Neuroimage 54, 2808–2821. doi: 10.1016/j.neuroimage.2010.10.069
Cutini, S., Basso Moro, S., and Bisconti, S. (2012). Functional near infrared optical imaging in cognitive neuroscience: an introductory review. J. Nearinfrared Spec. 20, 75–92. doi: 10.1255/jnirs.969
Daltrozzo, J., and Schön, D. (2009a). Conceptual processing in music as revealed by N400 effects on words and musical targets. J. Cogn. Neurosci. 21 1882–1892. doi: 10.1162/jocn.2009.21113
Daltrozzo, J., and Schön, D. (2009b). Is conceptual processing in music automatic? An electrophysiological approach. Brain Res. 1270 88–94. doi: 10.1016/j.brainres.2009.03.019
De Groot, A. (2006). Effects of stimulus characteristics and background music on foreign language vocabulary learning and forgetting. Lang. Learn. 56, 463–506. doi: 10.1111/j.1467-9922.2006.00374.x
Duncan, A., Meek, J. H., Clemence, M., Elwell, C. E., Fallon, P., Tyszczuk, L., et al. (1996). Measurement of cranial optical path length as a function of age using phase resolved near infrared spectroscopy. Pediatr. Res. 39 889–894. doi: 10.1203/00006450-199605000-00025
Eich, E. (1985). Context, memory, and integrated item/context imagery. J. Exp. Psychol. Learn. Mem. Cogn. 11, 764–770. doi: 10.1037/0278-7393.11.1-4.764
Ferrari, M., and Quaresima, V. (2012). A brief review on the history of human functional near-infrared spectroscopy (fNIRS) development and fields of application. Neuroimage 63 921–935. doi: 10.1016/j.neuroimage.2012.03.049
Ferreri, L., Bigand, E., Perrey, S., and Bugaiska, A. (in press). The promise of Near-Infrared Spectroscopy (NIRS) for psychological research: a brief review. Top Cogn. Psychol.
Foster, N. A., and Valentine, E. R. (2001). The effect of auditory stimulation on autobiographical recall in dementia. Exp. Aging Res. 27, 215–228. doi: 10.1080/036107301300208664
Franklin, M. S., Moore, K. S., Yip, C. Y., Jonides, J., Rattray, K., and Moher, J. (2008). The effects of musical training on verbal memory. Psychol. Music 36 353–365. doi: 10.1177/0305735607086044
Gagnon, L., Perdue, K., Greve, D. N., Goldenholz, D., Kaskhedikar, G., and Boas, D. (2011). Improved recovery of the hemodynamic response in diffuse optical imaging using short optode separations and state-space modeling. Neuroimage 56 1362–1371. doi: 10.1016/j.neuroimage.2011.03.001
Gagnon, L., Yücel, M. A., Dehaes, M., Cooper, R. J., Perdue, K. L., and Selb, J.et al. (2012). Quantification of the cortical contribution to the NIRS signal over the motor cortex using concurrent NIRS-fMRI measurements. Neuroimage 59, 3933–3940. doi: 10.1016/j.neuroimage.2011.10.054
Geday, J., and Gjedde, A. (2009). Attention, emotion, and deactivation of default activity in inferior medial prefrontal cortex. Brain Cogn. 69 344–352. doi: 10.1016/j.bandc.2008.08.009
Glisky, E. L., Polster, M. R., and Routhieaux, B. C. (1995). Double dissociation between item and source memory. Neuropsychology 9, 229–235. doi: 10.1037/0894-4105.9.2.229
Halpern, A. R. (2001). “Cerebral substrates of musical imagery,” in The Biological Foundations of Music, eds R. J. Zatorre and I. Peretz (New York: New York Academy of Science), 179–192.
Hamann, S. (2001). Cognitive and neural mechanisms of emotional memory. Trends. Cogn. Sci. (Regul. Ed.) 5 294–400. doi: 10.1016/S1364-6613(00)01707-1
Hertzog, C., Dunlosky, J., and Sinclair, S. (2010). Episodic feeling-of-knowing resolution derives from the quality of original encoding. Mem. Cognit. 38, 771–784. doi: 10.3758/MC.38.6.771
Ho, Y. C., Cheung M. C., and Chan, A. A. (2003). Music training improves verbal but not visual memory: cross-sectional and longitudinal explorations in children. Neuropsychology 17 439–450. doi: 10.1037/0894-4105.17.3.439
Hupbach, A., Hardt, O., Gomez, R., and Nadel, L. (2008). The dynamics of memory: context-dependent updating. Learn. Mem. 15, 574–579. doi: 10.1101/lm.1022308
Innocenti, I., Giovannelli, F., Cincotta, M., Feurra, M., Polizzotto, N. R., and Bianco,et al. (2010). Event-related rTMS at encoding affects differently deep and shallow memory traces. Neuroimage 53, 325–330. doi: 10.1016/j.neuroimage.2010.06.011
Janata, P., and Grafton, S. T. (2003). Swinging in the brain: shared neural substrates for behaviors related to sequencing and music. Nat. Neurosci. 6, 682–687. doi: 10.1038/nn1081
Janata, P., Tillmann, B., and Bharucha, J. J. (2002). Listening to polyphonic music recruits domain-general attention and working memory circuits. Cogn. Affect. Behav. Neurosci. 2, 121–140. doi: 10.3758/CABN.2.2.121
Jäncke, L., and Sandmann, P. (2010). Music listening while you learn: no influence of background music on verbal learning. Behav. Brain Funct. 6, 1–14. doi: 10.1186/1744-9081-6-3.
Jobsis, F. F. (1977). Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science 198, 1264–1267. doi: 10.1126/science.929199
Jurcak, V., Tsuzuki, D., and Dan, I. (2007). 10/20 10/10 and 10/5 systems revisited: their validity as relative head-surface-based positioning systems. Neuroimage 34, 1600–1611. doi: 10.1016/j.neuroimage.2006.09.024
Kensinger, E. A., and Corkin, S. (2003). Memory enhancement for emotional words: are emotional words more vividly remembered than neutral words? Mem. Cognit. 31, 1169–1180. doi: 10.3758/BF03195800
Kirilina, E., Jelzow, A., Heine, A., Niessing, M., Wabnitz, H., and Brühl, R.et al. (2012). The physiological origin of task-evoked systemic artefacts in functional near infrared spectroscopy. Neuroimage 61, 70–81. doi: 10.1016/j.neuroimage.2012.02.074
Koelsch, S., Kasper, E., Sammler, D., Schulze, K., Gunter, T., and Friederici, A. D. (2004). Music, language and meaning: brain signatures of semantic processing. Nat. Neurosci. 7, 302–307. doi: 10.1038/nn1197
Kohno, S., Miyai, I., Seiyama, A., Oda, I., Ishikawa, A., Tsuneishi, S.,et al. (2007). Removal of the skin blood flow artifact in functional near-infrared spectroscopic imaging data through independent component analysis. J. Biomed. Opt. 12, 062111 doi: 10.1117/1.2814249
Kroneisen, M., and Erdfelder, E. (2011). On the plasticify of the survival processing effect. J. Exp. Psychol. Learn. 37, 1553–1562. doi: 10.1037/a0024493
Kubota Y., Toichi, M., Shimizu, M., Mason, R. A., Findling, R. L., Yamamoto, K.,et al. (2006). Prefrontal hemodynamic activity predicts false memory – a near-infrared spectroscopy study. Neuroimage 31, 1783 – 1789. doi: 10.1016/j.neuroimage.2006.02.003
Lövdén, M., Rönnlund, M., and Nilsson, L. G. (2002). Remembering and knowing in adulthood: effects of enacted encoding and relations to processing speed. Aging Neuropsychol. 9, 184–200. doi: 10.1076/anec.9.3.184.9612
Maillet, D., and Rajah, M. N. (2012). Association between prefrontal activity and volume change in prefrontal and medial temporal lobes in aging and dementia: a review. Ageing Res. Rev. 12 479–489. doi: 10.1016/j.arr.2012.11.001
Manenti, R., Cotelli, M., Robertson, I. H., Miniussi., and C. (2012). Transcranial brain stimulation studies of episodic memory in young adults, elderly adults and individuals with memory dysfunction: a review. Brain Stimul. 5, 103–109. doi: 10.1016/j.brs.2012.03.004
Masataka, N., and Perlovsky, L. (2013). Cognitive interference can be mitigated by consonant music and facilitated by dissonant music. Sci. Rep. 3, 2028. doi: 10.1038/srep02028
Matsuda, G., and Hiraki, K. (2004). “Prefrontal cortex deactivation during video game play,” in Gaming. Simulation and Society: Research Scope and Perspective, eds R. Shiratori, K. Arai, and F. Kato (Springer-Verlag, Tokyo), 101–109.
Matsuda, G., and Hiraki, K. (2006). Sustained decrease in oxygenated hemoglobin during video games in the dorsal prefrontal cortex: a NIRS study of children. Neuroimage 29, 706–711. doi: 10.1016/j.neuroimage.2005.08.019
Matsui, M., Tanaka, K., Yonezawa, M., and Kurachi, M. (2007). Activation of the prefrontal cortex during memory learning: near-infrared spectroscopy study. Psychiatry Clin. Neurosci. 61, 31–38. doi: 10.1111/j.1440-1819.2007.01607.x
Mazoyer, P., Wicker, B., and Fonlupt, P. (2002). A neural network elicited by parametric manipulation of the attention load. Neuroreport 13, 2331–2334. doi: 10.1097/00001756-200212030-00032
Moghimi, S., Kushki, A., Guerguerian, A. M., and Chau T. (2012a). Characterizing emotional response to music in the prefrontal cortex using near infrared spectroscopy. Neurosci. Lett. 525 7–11. doi: 10.1016/j.neulet.2012.07.009
Moghimi S, Kushki A, Power S, Guerguerian AM, Chau T. (2012b). Automatic detection of a prefrontal cortical response to emotionally rated music using multi-channel near-infrared spectroscopy. J. Neural. Eng. 9, 1–8. doi: 10.1088/1741-2560/9/2/026022
Moussard, A., Bigand, E., Belleville, S., and Peretz, I. (2012). Music as an aid to learn new verbal information in Alzheimer’s disease. Music Percept. 29, 521–531. doi: 10.1525/mp.2012.29.5.521
Murray, L., J., and Ranganath, C. (2007). The dorsolateral prefrontal cortex contributes to successful relational memory encoding. J. Neurosci. 27 5515–5522. doi: 10.1523/JNEUROSCI.0406-07.2007
New, B., Pallier, C., Brysbaert, M., and Ferrand, L. (2004). Lexique 2: a new French lexical database. Behav. Res. Methods Instrum. Comput. 36, 516–524. doi: 10.3758/BF03195598
Nyberg, L., Cabeza, R., and Tulving, E. (1996). PET studies of encoding and retrieval: the HERA model. Psychon. Bull. Rev. 3, 135–148. doi: 10.3758/BF03212412
Okamoto, M., Dan, H., Sakamoto, K., Takeo, K., Shimizu, K., Kohno, S., andet al. (2004). Three-dimensional probabilistic anatomical cranio-cerebral correlation via the international 10–20 system oriented for transcranial functional brain mapping. Neuroimage 21, 99–111. doi: 10.1016/j.neuroimage.2003.08.026
Okamoto, M., Wada, Y., Yamaguchi, Y., Kyutoku, Y., Clowney, L., Singh, A. K., et al. (2011). Process-specific prefrontal contributions to episodic encoding and retrieval of tastes: a functional NIRS study. Neuroimage 54, 1578–1588. doi: 10.1016/j.neuroimage.2010.08.016
Parsons, L. M. (2001). Exploring the functional neuroanatomy of music performance, perception, and comprehension. Ann. N. Y. Acad. Sci. 930, 211–231. doi: 10.1111/j.1749-6632.2001.tb05735.x
Patel, A. D. (2011). Why would musical training benefit the neural encoding of speech? The OPERA hypothesis. Front. Psychol. 2:142. doi: 10.3389/fpsyg.2011.00142.
Peretz, I. (2002). Brain specialization for music. Neuroscientist 8, 372–380. doi: 10.1177/107385840200800412
Peterson, D. A., and Thaut, M. H. (2007). Music increases frontal EEG coherence during verbal learning. Neurosci. Lett. 412, 217–221. doi: 10.1016/j.neulet.2006.10.057
Prince, S. E., Daselaar, S. M., and Cabeza, R. (2005). Neural correlates of relational memory: successful encoding and retrieval of semantic and perceptual associations. J. Neurosci. 25, 1203–1210. doi: 10.1523/JNEUROSCI.2540-04.2005
Ptak, R. (2012). The frontoparietal attention network of the human brain: action, saliency, and a priority map of the environment. Neuroscientist 18, 502–515. doi: 10.1177/1073858411409051
Racette, A., Bard, C., and Peretz, I. (2006). Making non-fluent aphasics speak: sing along! Brain 129, 2571–2584. doi: 10.1093/brain/awl250
Racette, A., and Peretz, I. (2007). Learning lyrics: to sing or not to sing? Mem. Cognit. 35, 242–253. doi: 10.3758/BF03193445
Ramasubbu, R., Sing, H., Zhu, H., and Dunn, J. F. (2012). Methylphenidate-mediated reduction in prefrontal hemodynamic responses to working memory task: a functional near-infrared spectroscopy study. Hum. Psychopharm. Clin. 27, 615–621. doi: 10.1002/hup.2258
Ranganath, C. (2010). Binding items and contexts the cognitive neuroscience of episodic memory. Curr. Dir. Psychol. Sci. 19, 131–137. doi: 10.1177/0963721410368805
Salimpoor, V. N., Van Den Bosch, I., Kovacevic, N., McIntosh, A. R., Dagher, A., and Zatorre, R. J. (2013). Interactions between the nucleus accumbens and auditory cortices predict music reward value. Science 340, 216–219. doi: 10.1126/science.1231059
Särkämö, T., Tervaniemi, M., Latinen, S., Forsblom, A., Soinila, S., Mikkonen, M., et al. (2008). Music listening enhances cognitive recovery and mood after middle cerebral artery stroke. Brain 131, 866–876. doi: 10.1093/brain/awn013
Salthouse, T. A., Toth, J. P., Hancock, H. E., and Woodard, J. L. (1997). Controlled and automatic forms of memory and attention: process purity and the uniqueness of age-related influences. J. Gerontol. B Psychol. Sci. Soc. Sci. 52, 216–228. doi: 10.1093/geronb/52B.5.P216
Simmons-Stern, N. R., Budson, A. E., and Ally, B. A. (2010). Music as a memory enhancer in patients with Alzheimer’s disease. Neuropsychologia 48, 3164–3167. doi: 10.1016/j.neuropsychologia.2010.04.033
Schellenberg, E. G. (2003). “Does exposure to music have beneficial side effects?” in The Cognitive Neuroscience of Music, eds I. Peretz and R. J. Zatorre (Oxford, NY: Oxford University Press), 430–444. doi: 10.1093/acprof:oso/9780198525202.003.0028
Schellenberg, E. G. (2005). Music and cognitive abilities. Curr. Dir. Psychol. Sci. 14 317–320. doi: 10.1111/j.0963-7214.2005.00389.x
Spaniol, J., Davidson, P. S. R., Kim, A., Han, H., Moscovitch, M., and Grady, C. L. (2009). Event-related fMRI studies of episodic encoding and retrieval: meta-analyses using activation likelihood estimation. Neuropsychologia 47, 1765–1779. doi: 10.1016/j.neuropsychologia.2009.02.028
Spector, A. and Biederman, I. (1976). Mental set and mental shift revisited. Am. J. Psychol. 89 669–679. doi: 10.2307/1421465
Takahashi, T., Takikawa, Y., Kawagoe, R., Shibuya, S., Iwano, T., and Kitazawa, S. (2011). Influence of skin blood flow on near-infrared spectroscopy signals measured on the forehead during a verbal fluency task. Neuroimage 57 991–1002. doi: 10.1016/j.neuroimage.2011.05.012
Thaut, M. H., Peterson, D. A., and McIntosh, G. C. (2005). Temporal entrainment of cognitive functions musical mnemonics induce brain plasticity and oscillatory synchrony in neural networks underlying memory. Ann. N. Y. Acad. Sci. 1060, 243–254. doi: 10.1196/annals.1360.017
Thompson, R. G., Moulin, C. J. A., Hayre, S., Jones, R. W. (2005). Music enhances category fluency in healthy older adults and Alzheimer’s disease patients. Exp. Aging Res. 31 91–99. doi: 10.1080/03610730590882819
Tulving, E. (1972). “Episodic and semantic memory,” in Organization of Memory, eds E. Tulving and W. Donaldson (New York: Academic Press), 381–403
Tulving, E., Kapur, S., Craik, F. I. M., Moscovitch, M., and Houle, S. (1994). Hemispheric encoding/retrieval asymmetry in episodic memory: positron emission tomography findings. Proc. Natl. Acad. Sci. U.S.A. 91, 2016–2020. doi: 10.1073/pnas.91.6.2016
Wallace, W. T. (1994). Memory for music: effect of melody on recall of text. J. Exp. Psychol. Learn. 20, 1471–1485. doi: 10.1037/0278-7393.20.6.1471
Keywords: music, verbal memory, encoding, prefrontal cortex, fNIRS
Citation: Ferreri L, Aucouturier J-J, Muthalib M, Bigand E and Bugaiska A (2013) Music improves verbal memory encoding while decreasing prefrontal cortex activity: an fNIRS study. Front. Hum. Neurosci. 7:779. doi: 10.3389/fnhum.2013.00779
Received: 30 September 2013; Paper pending published: 06 October 2013;
Accepted: 29 October 2013; Published online: 22 November 2013.
Edited by:
Nobuo Masataka, Kyoto University, JapanReviewed by:
Leonid Perlovsky, Harvard University; Air Force Research Laboratory, USACopyright © 2013 Ferreri, Aucouturier, Muthalib, Bigand and Bugaiska. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Laura Ferreri, Laboratory for the Study of Learning and Development, CNRS UMR 5022, Department of Psychology, University of Burgundy, Pôle AAFE-11 Esplanade Erasme, 21000 Dijon, France e-mail:lf.ferreri@gmail.com
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.