- Division of Electrophysiology, Department of Cardiology, University of California San Francisco, San Francisco, CA, USA
Background
Arrhythmogenic right ventricular dysplasia/cardiomyopathy (ARVD/C) is an inherited disease characterized by a progressive replacement of myocytes with fibrofatty tissue. The replacement primarily affects the right ventricle but can progress to affect the left ventricle as well (Marcus et al., 1982; Thiene et al., 1988; Marcus and Fontaine, 1995; Basso et al., 2009). The progressive spread starts in the epicardium and eventually becomes transmural but with sparing of the septum.
Epidemiology of Ventricular Arrhythmias
Most patients with ARVD/C come to medical attention after presenting with ventricular arrhythmias. This is in agreement with the initial series of ARVD/C cases first reported by Marcus et al. (1982) reporting 24 patients all presenting with ventricular arrhythmias. However more recently registry data allows insight into less advanced presentation of disease. In the North American Multidisciplinary ARVD study, patient characteristics were classified in 108 probands with suspected ARVD/C (Marcus et al., 2009). Ventricular arrhythmias were not identified in every patient. Sustained clinical ventricular tachycardia (VT) was present in 35% and among patients undergoing electrophysiology study (75 probands) while sustained VT was induced during an electrophysiology study in 49%. Among the patients with sustained VT, the morphology of LBBB/superior axis was the most common (14/38, 36.8%), LBBB/inferior axis (10/38, 26.3%), with the remaining having either LBBB/indeterminate axis (8/38, 21%), indeterminate morphology (5/38, 13%), and RBBB pattern (1/38, 2.6%).
Electrocardiographic Differentiation between RVOT-VT and ARVD/C
Differentiation between idiopathic VT from the right ventricular outflow tract (RVOT-VT) and ARVD/C is of utmost importance given the benign nature of the former and the need for sudden cardiac risk stratification and family screening in the latter (Calkins, 2006). Baseline sinus rhythm electrocardiography as well as electrocardiographic differences during ventricular arrhythmias (VT or PVCs) can be helpful in differentiating the two disease states.
Sinus Rhythm
Baseline sinus rhythm electrocardiography may be useful. The presence of T wave inversion in V1–V3 in baseline sinus rhythm may aid the diagnosis of ARVD/C, but recent data have shown that these changes may be present in only 32% of ARVD/C patients as well as 1–3% of normal young patients and 4% of RVOT-VT patients (Kazmierczak et al., 1998; Marcus, 2005; Marcus et al., 2009; Morin et al., 2010). In fact in an early series comparing ECG characteristics between ARVD/C and RVOT-VT, Kazmierczak et al. (1998) found anterior T wave inversion in 50% of the ARVD/C group and 20% in their RVOT-VT group. Morin et al. (2010) evaluated the testing characteristics of anterior T wave inversions (V1–V3) in 79 patients with ARVD/C, and in 121 patients with RVOT-VT. During sinus rhythm, 37 patients (47%) with ARVC and 5 patients (4%) with RVOT tachycardia had anterior T wave inversions, with resultant sensitivity of 47% and specificity of 96% for the diagnosis of ARVD/C.
Epsilon waves, the pathognomonic low-amplitude signals between the end of QRS complex to onset of the T wave in the right precordial leads (V1–V3) if seen is very helpful and is a major diagnostic criterium (Marcus et al., 2010), but is seen in the minority of patient in most series, 10–37% (Kazmierczak et al., 1998; Arbelo and Josephson, 2010). In fact in our series not a single ARVD/C patient had epsilon waves (Hoffmayer et al., 2011).
Ventricular Arrhythmias
Single versus Multiple Forms
The presence of more than one form of VT should increase the pre-test probability for ARVD/C as this likely reflects more diffuse pathology affecting multiple sites of the right ventricle. However the predictive value of this finding is not 100% accurate. Early in the disease of ARVD/C one may see a single form of VT. There has also been a report of multiple forms of VT seen in an idiopathic VT, but the diagnosis in these rare instances must be carefully scrutinized.
Niroomand et al. (2002) performed electrophysiology studies in 56 patients with ventricular arrhythmias, based on idiopathic RVOT-VT in 41 and ARVD/C in 15. The presence of more than one type of VT was seen in 75% of the ARVD group compared with 0% in the idiopathic RVOT-VT group, p < 0.0001). The electrophysiology study was also helpful in differentiating the two groups, as inducibility of VT by programmed electrical stimulation with ventricular extrastimuli was seen in 93% of the ARVD/C group compared with only 3% of the RVOT-VT group, p < 0.0001. Fragmented diastolic potentials during ventricular arrhythmia was seen in 93% of the RVOT-VT group compared to 0% in the RVOT-VT group, p < 0.0001.
O’Donnell et al. (2003) performed electrophysiology studies in 50 patients, with 33 RVOT-VT and 17 patients having ARVD/C. The presence of more than one type of VT morphology was seen in 71% of the ARVD/C group and 0% in the RVOT-VT group, p < 0.01.
Kazmierczak et al. (1998) in the mid 1990s evaluated ECG characteristics in 15 patients with repetitive monomorphic VT (RMVT) of RV origin compared with 12 ARVD cases. One of the 15 cases of RMVT had multiple VT patterns, although the exact number and type were not described. This was the same study that showed baseline anterior T wave changes in 20% of the RVOT-VT group. This observation is at variance with most reports.
VT/PVC Morphology
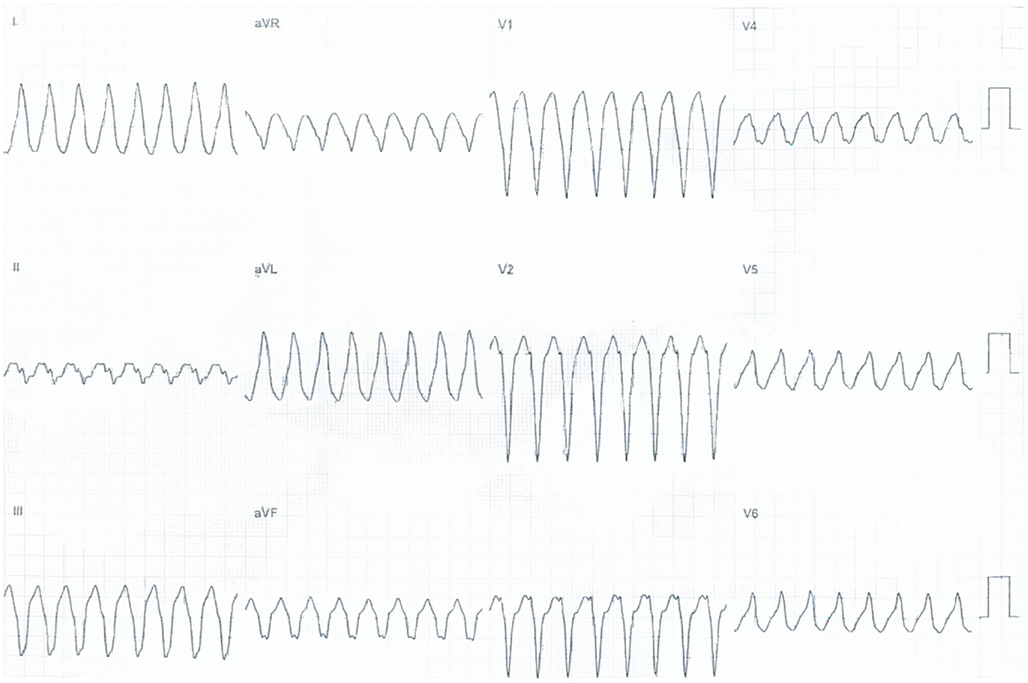
The electrocardiographic morphology of the VT/PVCs has been used to differentiate the two entities. Multiple VT forms including LBBB/superior axis essentially excludes RVOT-VT and should shift your pre-test probability toward ARVD/C (Arbelo and Josephson, 2010). An example of LBBB/superior axis VT from an ARVD/C patient is shown in Figure 1.
Ainsworth et al. (2006) analyzed QRS duration to help differentiate ARVD/C from RVOT-VT. They found that mean QRS duration was longer in all 12 leads in ARVC patients with a significant difference seen in leads I, III, aVL, aVF, V1, V2, and V3 (p < 0.05). Leads I and aVL had the largest mean difference between ARVC and RVOT-VT patients of 17.6 ± 4.7 and 15.8 ± 7.5 ms, respectively (p = 0.0001). Lead I QRS duration ≥120 ms had a sensitivity of 100%, specificity 46%, positive predictive value 61%, and negative predictive value (NPV) 100% for ARVC.
More recently we found several ECG criteria helpful in distinguishing between ARVD/C and RVOT-VT (Hoffmayer et al., 2011). ARVD/C patients had a significantly longer mean QRS duration in lead I (150 ± 31 versus 123 ± 34 ms, p = 0.006), more often exhibited a precordial transition in lead V6 [3/17 (18%) versus 0/42 (0%) with RVOT-VT, p = 0.005] and more often had at least one lead with notching [11/17 (65%) versus 9/42 (21%), p = 0.001]. The most sensitive characteristics for the detection of ARVD/C was a QRS duration in lead I of ≥120 ms (sensitivity 88%, NPV 91%). QRS transition at V6 was most specific 100% (PPV 100%, NPV 77%). In multivariate analysis, QRS duration in lead I ≥ 120 ms (OR 20.4, p = 0.034), earliest onset QRS in lead V1 (OR 17.0, p = 0.022), QRS notching (OR 7.7, p = 0.018), and a transition V5 or later (OR 7.0, p = 0.030) each predicted an increased odds of ARVD/C. Figure 2 shows examples of these findings.
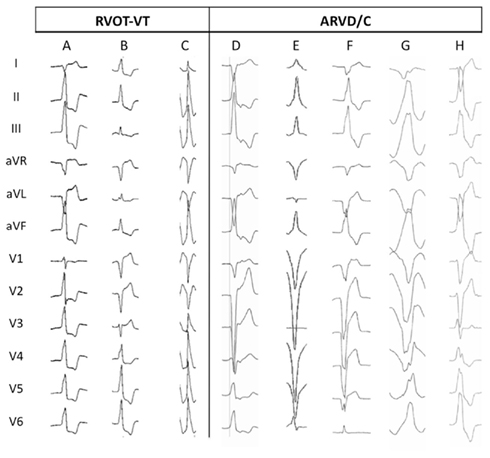
Figure 2. Ventricular tachycardia morphology characteristics (Hoffmayer et al., 2011). Twelve lead ECGs from patients with RVOT-VT (A–C) and ARVD/C (D–H) showing characteristic features. (A) RVOT-VT from anterior–septal location, showing precordial transition at V2, and narrow QRS duration in lead I (78 ms). (B) RVOT-VT originating superior to his bundle region, showing precordial transition at V4, positive R wave in aVL and narrow QRS in lead I (86 ms). (C) RVOT-VT from posterior–septal location, showing precordial transition at V3, and narrow QRS duration in lead I (118 ms). (D) ARVD/C VT shows late precordial transition V5, wide QRS duration in lead I (124 ms), and earliest onset QRS in V1 (vertical line). (E) ARVD/C VT shows very late precordial transition V6 and wide QRS duration in lead I (126 ms). (F) ARVD/C VT shows very late precordial transition V6 and wide QRS duration in lead I (150 ms). (G) ARVD/C VT shows late precordial transition V5, wide QRS duration in lead I (160 ms), and notching of the QRS (II, III, aVF, V4–6). (H) ARVD/C VT shows wide QRS duration in lead I (128 ms) and notching of the QRS (II, III, aVF, V4–6).
Pathophysiology of Ventricular Tachycardia Morphology
The mechanism of the differences in QRS morphology in ARVD/C compared to RVOT-VT is related to the difference in underlying pathology in the two conditions. The underlying pathophysiologic mechanism in ARVD/C is replacement of normal right ventricular myocardial tissue with fibrous and fatty tissue. This replacement may delay cell to cell conduction and facilitate the development of reentrant ventricular arrhythmias (Thiene et al., 1988). This results in a greater delay from earliest onset to local onset of the QRS complex, greater duration of the QRS complex, and irregularities of conduction manifest as notching of the QRS complex.
Another factor may be related to VT anatomical location. ARVD/C patients have a greater frequency of the site of origin of the right ventricular free-wall, more remote from the normal His–Purkinje conduction tissue than in those patients with RVOT-VT. These factors also explain the late precordial transition and possibly notching of the QRS. A number of studies have found that VT origins in patients with RVOT-VT are predominately in the septum (Dixit et al., 2003; Joshi and Wilber, 2005). Therefore, differences between ECG characteristics of ventricular arrhythmias in patients with RVOT-VT versus ARVD/C are partially explained by differences in the site of VT origin.
Summary
Ventricular arrhythmias in patients with ARVD/C are common. Differentiation between idiopathic VT and ARVD is of utmost importance. Baseline sinus rhythm electrocardiography as well as electrocardiographic differences during ventricular arrhythmias (VT or PVCs) can be helpful in differentiating the two disease states. Anterior T wave inversions in sinus rhythm, multiple VT forms, wider QRS duration (lead I ≥ 120 ms), QRS notching, earliest onset QRS in lead V1 later, and precordial transition (>V5) all increase the odds of ARVD/C.
References
Ainsworth, C. D., Skanes, A. C., Klein, G. J., Gula, L. J., Yee, R., and Krahn, A. D. (2006). Differentiating arrhythmogenic right ventricular cardiomyopathy from right ventricular outflow tract ventricular tachycardia using multilead QRS duration and axis. Heart Rhythm 3, 416–423.
Arbelo, E., and Josephson, M. E. (2010). Ablation of ventricular arrhythmias in arrhythmogenic right ventricular dysplasia. J. Cardiovasc. Electrophysiol. 21, 473–486.
Basso, C., Corrado, D., Marcus, F. I., Nava, A., and Thiene, G. (2009). Arrhythmogenic right ventricular cardiomyopathy. Lancet 373, 1289–1300.
Calkins, H. (2006). Arrhythmogenic right-ventricular dysplasia/cardiomyopathy. Curr. Opin. Cardiol. 21, 55–63.
Dixit, S., Gerstenfeld, E. P., Callans, D. J., and Marchlinski, F. E. (2003). Electrocardiographic patterns of superior right ventricular outflow tract tachycardias: distinguishing septal and free-wall sites of origin. J. Cardiovasc. Electrophysiol. 14, 1–7.
Hoffmayer, K. S., Machado, O. N., Marcus, G. M., Yang, Y., Johnson, C. J., Ermakov, S., Vittinghoff, E., Pandurangi, U., Calkins, H., Cannom, D., Gear, K. C., Tichnell, C., Park, Y., Zareba, W., Marcus, F. I., and Scheinman, M. M. (2011). Electrocardiographic comparison of ventricular arrhythmias in patients with arrhythmogenic right ventricular cardiomyopathy and right ventricular outflow tract tachycardia. J. Am. Coll. Cardiol. 58, 831–838.
Joshi, S., and Wilber, D. J. (2005). Ablation of idiopathic right ventricular outflow tract tachycardia: current perspectives. J. Cardiovasc. Electrophysiol. 16(Suppl. 1), S52–S58.
Kazmierczak, J., De Sutter, J., Tavernier, R., Cuvelier, C., Dimmer, C., and Jordaens, L. (1998). Electrocardiographic and morphometric features in patients with ventricular tachycardia of right ventricular origin. Heart 79, 388–393.
Marcus, F. I. (2005). Prevalence of T-wave inversion beyond V1 in young normal individuals and usefulness for the diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia. Am. J. Cardiol. 95, 1070–1071.
Marcus, F. I., and Fontaine, G. (1995). Arrhythmogenic right ventricular dysplasia/cardiomyopathy: a review. Pacing Clin. Electrophysiol. 18, 1298–1314.
Marcus, F. I., Fontaine, G. H., Guiraudon, G., Frank, R., Laurenceau, J. L., Malergue, C., and Grosgogeat, Y. (1982). Right ventricular dysplasia: a report of 24 adult cases. Circulation 65, 384–398.
Marcus, F. I., McKenna, W. J., Sherrill, D., Basso, C., Bauce, B., Bluemke, D. A., Calkins, H., Corrado, D., Cox, M. G., Daubert, J. P., Fontaine, G., Gear, K., Hauer, R., Nava, A., Picard, M. H., Protonotarios, N., Saffitz, J. E., Sanborn, D. M., Steinberg, J. S., Tandri, H., Thiene, G., Towbin, J. A., Tsatsopoulou, A., Wichter, T., and Zareba, W. (2010). Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the task force criteria. Circulation 121, 1533–1541.
Marcus, F. I., Zareba, W., Calkins, H., Towbin, J. A., Basso, C., Bluemke, D. A., Estes, N. A. III., Picard, M. H., Sanborn, D., Thiene, G., Wichter, T., Cannom, D., Wilber, D. J., Scheinman, M., Duff, H., Daubert, J., Talajic, M., Krahn, A., Sweeney, M., Garan, H., Sakaguchi, S., Lerman, B. B., Kerr, C., Kron, J., Steinberg, J. S., Sherrill, D., Gear, K., Brown, M., Severski, P., Polonsky, S., and McNitt, S. (2009). Arrhythmogenic right ventricular cardiomyopathy/dysplasia clinical presentation and diagnostic evaluation: results from the North American Multidisciplinary Study. Heart Rhythm 6, 984–992.
Morin, D. P., Mauer, A. C., Gear, K., Zareba, W., Markowitz, S. M., Marcus, F. I., and Lerman, B. B. (2010). Usefulness of precordial T-wave inversion to distinguish arrhythmogenic right ventricular cardiomyopathy from idiopathic ventricular tachycardia arising from the right ventricular outflow tract. Am. J. Cardiol. 105, 1821–1824.
Niroomand, F., Carbucicchio, C., Tondo, C., Riva, S., Fassini, G., Apostolo, A., Trevisi, N., and Bella, P. D. (2002). Electrophysiological characteristics and outcome in patients with idiopathic right ventricular arrhythmia compared with arrhythmogenic right ventricular dysplasia. Heart 87, 41–47.
O’Donnell, D., Cox, D., Bourke, J., Mitchell, L., and Furniss, S. (2003). Clinical and electrophysiological differences between patients with arrhythmogenic right ventricular dysplasia and right ventricular outflow tract tachycardia. Eur. Heart J. 24, 801–810.
Citation: Hoffmayer K and Scheinman MM (2012) Electrocardiographic patterns of ventricular arrhythmias in arrhythmogenic right ventricular dysplasia/cardiomyopathy. Front. Physio. 3:23. doi: 10.3389/fphys.2012.00023
Received: 16 December 2011;
Accepted: 27 January 2012;
Published online: 16 February 2012.
Copyright: © Hoffmayer and Scheinman. This is an open-access article distributed under the terms of the Creative Commons Attribution Non Commercial License, which permits non-commercial use, distribution, and reproduction in other forums, provided the original authors and source are credited.
*Correspondence: scheinman@medicine.ucsf.edu