Rapid Characterization of Microcystin-Producing Cyanobacteria in Freshwater Lakes by TSA-FISH (Tyramid Signal Amplification-Fluorescent In Situ Hybridization)
- 1UMR 6553 Centre National de la Recherche Scientifique ECOBIO/OSUR, University of Rennes 1, Rennes, France
- 2Aix Marseille University, CNRS, IRD, University of Toulon, Mediterranean Institute of Oceanography, UM110, Marseille, France
- 3Institut National de la Santé et de la Recherche Médicale U1193, Physiopathogénèse et Traitement des Maladies du Foie, Hôpital Paul Brousse, Villejuif, France
- 4LIED, UMR 8236 Centre National de la Recherche Scientifique, University Paris-Diderot, Paris, France
- 5Chimie ParisTech, ENSCP, Paris, France
Microcystin (MC) is a common and widespread toxin which represents a health hazard to humans and animals. MC toxin concentrations are monitored by various direct or proxy techniques (HPLC, LC-MS/MS, ELISA, PPIA), however, these techniques do not discriminate producing species from non-producing ones. In order to simultaneously provide the identity and activity of cyanotoxin producing species in freshwater lakes, we applied simple, and fully detailed, whole cell fluorescent in situ hybridization enhanced by tyramid signal amplification (TSA-FISH). DNA oligonucleotide probes MICR3 and MCYA were targeting 16S rRNA and mcyA-mRNA, respectively. The mcyA gene is coding for the MC synthetase enzyme involved in MC synthesis. Controls were acquired with the general eubacterial 16S rRNA probe EUB338, for TSA-FISH assay, and standard HPLC and LC-MS/MS as standard methods for the measurements of MC concentration. Results obtained from monoclonal strains and natural samples demonstrated a specific identification of Microcystis species and were able to discriminate MC producing from non-producing ones. In addition, the MCYA probe allowed the specific detection of MC-synthetase mRNA within Planktothrix isothrix (Oscillatoriale) filaments. Two kinds of mcyA-mRNA labeling were observed in these cells, spots like and plasmid like, which illustrates the well-known plasticity of microbial genome to adapt to environmental stresses. We demonstrated that a simple TSA-FISH assay allows acquiring rapidly dual information of the presence and abundance of potentially toxic species, while identifying species actively producing MC-synthetase mRNA, a proxy of MC toxin. This technique has the potential to be developed into an effective environmental monitoring tool. In addition, detail visualization of cellular mRNAs is powerful for the acquisition of ecological and biomolecular studies of toxic cyanobacteria.
Introduction
Many freshwater cyanobacteria produce blooms which may be toxic to fauna, flora, and humans (Chorus and Bartram, 1999) and raises the importance of making rapid diagnosis of toxin content and/or the toxic potential of these blooms. General phycocyanin probes can be easily deployed in a natural environment to rapidly detect total cyanobacteria concentrations (Brient et al., 2008), while toxin concentrations are measured by standard HPLC or mass spectrometry based techniques (Lawton and Edwards, 2008). Although these latter methods are time consuming, they remained references for the calibration of rapid biochemical (ELISA and PPIA) assays. Potentially toxic species are often identified by biomolecular (PCR) assays (Carmichael and An, 1999; Pearson and Neilan, 2008; Humpage et al., 2012) but they involve DNA extraction which destroys cells and colony organization. Identification of cyanotoxin producing species requires additional time consuming microscopic isolation, purification, and cultivation. Thus a rapid assay, which simultaneously provides the identity and the activity of cyanotoxin producing species, still remains a challenge.
The mcyA gene has been frequently used in many molecular based approaches, as it is involved in the ribosomal production of microcystine (MC) synthetase, responsible for MC production, through non-ribosomal pathways (Tillett et al., 2000). Many studies have shown a transcriptional regulation of mcyA gene in accordance with changes in toxin concentrations, although discrepancies have been described since early studies which suggested a post-transcriptional regulation of mcyA gene expression (Schawbe et al., 1988; Meissner et al., 1996). Very recently, a whole cell Tyramid Signal Amplification-Fluorescent in situ Hybridization (TSA-FISH) assay was developed to target mcyA mRNA transcription as a proxy of MC-synthetase production (Zeller et al., 2016). This assay could verify an up-regulation of mcyA-mRNA synthesis by light, and show similar spot-like fluorescent labeling as the rpob-mRNA probe, where the rpob gene is involved in RNA polymerase synthesis (Gaget et al., 2011). That study demonstrated the potential interest to distinguish toxic from non-toxic organisms, by combining the precision of molecular based technique with species identification, as cellular integrity was not destroyed. However, direct measurement of MC toxin by standard HPLC methods was not provided, and it's in situ application remained to be demonstrated.
The aim of the present study was to demonstrate that (i) mcyA-mRNA TSA-FISH assay could label MC producing laboratory strains, as well as cells from the natural environment and (ii) identification of MC producing species could be easily acquired simultaneously by the use of specific identity MICR3-16S rDNA probe; when (iii) toxin concentrations were verified by standard HPLC and/or LC-MS/MS protocols.
Methods
Samples
Two non-axenic cultures of Microcystis aeruginosa from the Pasteur Culture Collection (PCC, France) were used: the PCC 7806 microcystin MC-producing and a non-MC-producing PCC 9432. Two Oscillatoriales strains were also used: Planktothrix rubescens UR1 PR1 (University of Rennes 1) a MC-producing strain, isolated in our laboratory according to Shirai et al. (1989, 1991), and the non-toxic Planktothrix agardhii PCC 7805. They were grown in BG11 medium at 20 ± 1°C with a 14 h/10 h of L/D period, with 38 μmol photons m−2 s−1. Lake samples were from Vioreau reservoir (Loire-Atlantic, France) and the Drennec Lake (Brittany, France).
Microcystins Analysis
Microcystins were extracted from strains and natural samples after sample filtration in 75% methanol (HiperSolv CHROMANORN) according to Meriluoto and Codd (2005) and Spoof et al. (2003). The toxin was then identified and quantified using high-performance liquid chromatography (HPLC, SpectraSYSTEM P4000), followed by UV absorbance diode-array detection (DAD, SpectraSYSTEM UV6000) or mass spectrometry LC-MS/MS for Drennec lake samples for validation of desmethyl RR-microcystin.
Molecular Probes and Phylogenetic Specificity
Oligonucleotide probes used for TSA-FISH assays were obtained from the literature: the EUB338 probe (5′-GCT GCC TCC CGT AGG AGT-3′) targets a part of the 16S rRNA Eubacteria domain (Amann et al., 1990); the MICR3 probe (5′-TCT GCC AGT TTC CAC CGC CTT TAG GT-3′) is specific for 16S rRNA Microcystis spp. (Rudi et al., 2000); and the MCYA probe (5′-ATG AGC CGC CAA TAA AAC ACT TT-3′) is specific for mcyA mRNA of MC-synthetase (Hisbergues et al., 2003; Zeller et al., 2016). All probes were purchased with a 3′-C6 amino link conjugated to horseradish peroxidase (HRP) to ensure TSA amplification, according to Urdea et al. (1988) modification (Thermo-Fisher Scientific, Germany). Specificity of MCYA TSA-FISH probe was confirmed (Zeller et al., 2016), while specificity of MICR3 probe was verified by in silico analysis through the Multiple Sequence Alignment (MSA) of the on-line ClustalW2 software (EMBL-EBI, Cambridge, http://www.ebi.ac.uk/). The alignment matched with the 16S rRNA sequence of M. aeruginosa PCC7806 (accession number: U03402.1), and showed four mismatches with Woronichinia sp., the closest non-targeted phylogenetic sequence (accession number: JN172622.1; 5′-GGT CTC AGT TTC CAC CGC CTT TAT GG-3′, bold bases are mismatches with MICR3 probe). Microcystis and Woronichinia genera were used as positive and negative controls, respectively, for MICR3 probe either in cultures or in the field.
TSA-FISH and Microscopic Observations
TSA-FISH assays were produced according to Biegala et al. (2002); Biegala and Raimbault (2008) with slight modifications, including DAPI (4′,6-diamidine-2′-phenylindole dihydrochloride) counterstaining, which was replaced by propidium iodide (PI) (Biegala et al., 2003). Protocol steps are detailed in Table S1 and information on equipment and solutions are provided in Table S2. Cyanobacterial autofluorescence from cultures or natural environment did not prevent from FITC signal observation, thus copper sulfate treatment proposed previously (Zeller et al., 2016) was not applied in thisstudy.
Micrographs were performed with an epifluorescence microscope (OLYMPUS BX41) equipped with a 100 W short arc HBO mercury lamp and dichroïc filters 460–490 and 520 nm em. LP (long pass) for FITC; and 510–550 and 590 nm em. LP for PI. Photomicrographs were captured by a camera (Leica DMC 2900) controlled by Leica Application Suite (LAS) V4.5. Pictures were acquired with specific time exposures set on the positive control and kept constant on negative control according to Riou et al. (2017). Once acquired, pictures were not modified except for PI to provide better pictures.
Results and Discussion
In this study, we successfully applied a simple TSA-FISH protocol (Table S1) to detect specifically in cultures and the natural environment MC-producing cells. Positive control of TSA-FISH assay, both in culture and the natural environment, was ensured by the eubacterial domain specific probe EUB338 (Figures 1, 2) which brightly labeled cells with fluorescein fluorochrome (FITC). Presence of Microcystis genus was specifically targeted by the MICR3 probe (Figures 1, 2B1,B2, 3A, 4A,B) while the Chroococcale Woronichinia spp., the closest phylogenetic none specific genus, were not targeted (Figure 4A thin arrow, Figure 4B thick arrow), nor the most distantly related filamentous Nostocale Dolichospermum mendotae (Figures 2B1,B2 arrow). TSA-FISH assay is a widely used technique for the specific detection and quantification of cyanobacteria (Schonhuber et al., 1999; West et al., 2001; Biegala and Raimbault, 2008). However, the detection of messenger RNA has long been believed to be challenging (Pernthaler and Amann, 2004) because of low copy number of specific mRNA copies per cell. Trcek et al. (2012) and more recently (Zeller et al., 2016) successfully labeled single mRNA within microorganisms, including M. aeruginosa strains. Both studies showed the precision of in situ hybridization approaches using either multiple monolabeled DNA probe (DIG-FISH) or a simple TSA-FISH assay, respectively. In addition, once fixed with PFA (paraformaldehyde) all proteins are crosslinked which prevent RNA degradation from endogenous RNAse. Both observations justified the use in this study of similar TSA-FISH protocol to target whole cell rRNA and mRNA.
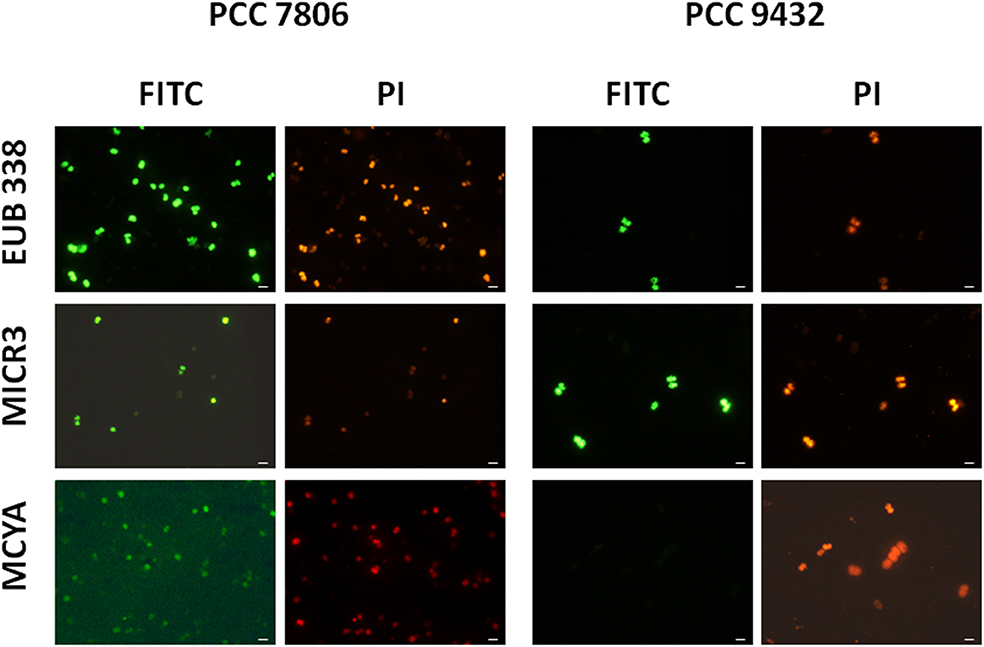
Figure 1. Micrographs showing the validation of the Microcystis spp. specific MICR3 probe and the MC-synthetase specific MCYA probes on Microcystis aeruginosa PCC 7806 (MC-producing) and PCC 9432 (non-MC-producing, Table 1) strains. The EUB338 domain specific probe was used as a general positive control. Green and red fluorescences show FITC labeled probe and PI-linked-DNA dyes, respectively. Exposure = 150 ms. Scale bar = 10 μm.
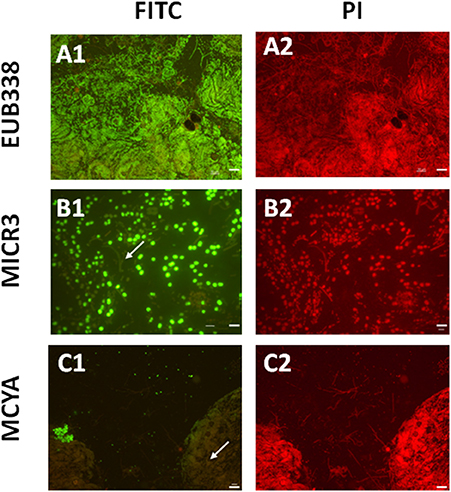
Figure 2. Micrographs showing in situ the validation of the Microcystis spp. specific MICR3 probe and the MC-synthetase specific MCYA probes on natural samples from Vioreau lake containing a mix of cyanobacteria and eukaryotes. EUB388 probe is a positive control of hybridization (A1,A2). Only Microcystis aeruginosa were targeted by MICR3 probe, while the filamentous Dolichospermium mendotae was not labeled (arrow B1,B2). Only microcystin producing cells (Microcystis viridis) were hybridized by MCYA probe, while Microcystis flos aquae was not labeled (arrow C1,C2, Table 1). Arrows show non labeled species. Green and red fluorescence show FITC labeled probe and PI-linked-DNA dyes, respectively. Exposure = 400 ms. Scale bars = 50 μm (A1,A2,C1,C2), and = 10 μm (B1,B2).
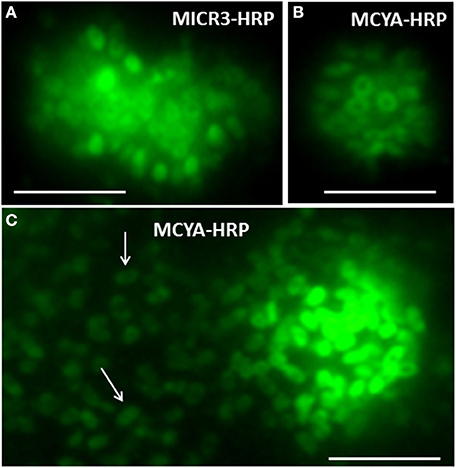
Figure 3. Micrographs of toxic Microcystis aeruginosa 7806 strains showing rRNA and mRNA labeled, respectively, with MICR3 (A) and MCYA (B,C) probes in the periphery of each plasmid. Arrows point to extra cellular plasmids released by the cell. Green is FITC probe labeling by TSA-FISH Exposure is 900 nm. Scale Bar = 5 μm.
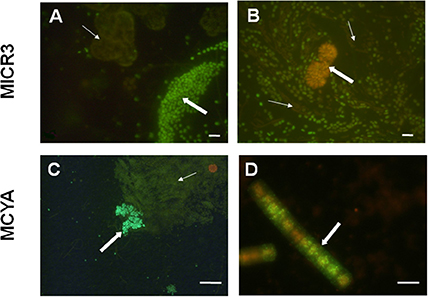
Figure 4. Micrographs showing in situ specific detection of Microcystis spp. and microcystin producers from Vioreau reservoir and Drennec lake. Panel (A) shows the Microcystis wesenbergii (thick arrow) targeted by MICR3 probe and the non-labeled Woronichinia naegeliana (fine arrow) which holds four mismatches with the MICR3 probe (negative control). Panel (B) shows Microcystis spp. labeled with MICR3 probe, while the filamentous Dolichospermum mendotae (fine arrow) and the densed colony forming Woronichinia naegeliana (thick arrow, negative control) were not labeled. Panel (C) shows Microcystis viridis (thick arrow) labeled with MCYA probe, when the non-producing Microcystis flos aquae (fine arrow) and the Woronichinia naegeliana (orange rounded colony, control) were not labeled by the probe. Panel (D) shows the microcystin producing Planktothrix isothrix from Drennec lake, labeled with MCYA probe. Exposure = 400 ms (A–C), = 500 ms (D) Scale bar = 20 μm (A), = 100 μm (B), = 50 μm (C), = 5 μm (D).
We further demonstrated in this study that MCYA probe, specific for MC-synthetase messenger RNA, only targeted toxic Chrooccocales cells belonging to three different species of Microcystis. These species are M. aeruginosa from cultivated strains (Figure 1, Table 1), Microcystis viridis and Microcystis wesenbergii from Vioreau reservoir (Figures 2C1, 4C, Table 1). On the contrary, the non-MC-producing M. aeruginosa PCC 9432 strain (Figure 1, Table 1) and the Microcystis flos aquae from Vioreau reservoir were not targeted (Figure 2C1 arrow, Table 1). In addition, the MCYA probe labeled mcyA mRNA as many small spots within P. rubescens toxic strain (Figure 5, Table 1), as well as the highly toxic bloom of Planktothrix isothrix from Drennec lake (Figure 4D arrow, Table 1), while filaments from the non-MC-producing P. agardhii PCC 7805 were not labeled by MCYA (Figure 5, Table 1). Unexplained uniform green fluorescence could also be seen in some filaments from this non-toxic PCC 7805 strain.
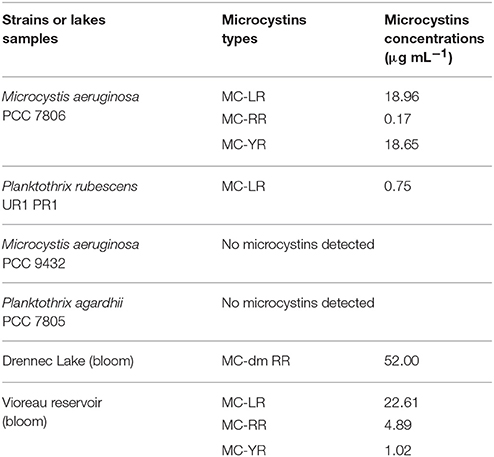
Table 1. Microcystins types and concentrations in cultivated strains and environmental freshwater samples.
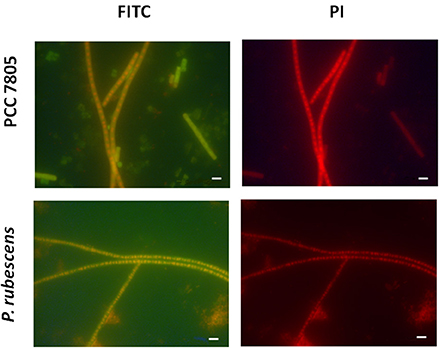
Figure 5. Micrographs showing the validation of MC-synthetase specific MCYA probes on cultures of filamentous Oscillatoriale Planktothrix agardhii PCC 7805 (non-MC-producing) and P. rubescens (Table 1, MC-producing). Green and red fluorescence show FITC labeled probe and PI-linked-DNA dyes, respectively. Exposure = 400 ms. Scale bar = 10 μm.
According to our results and those from Zeller et al. (2016) it is interesting to note that two kinds of intracellular labeling could be observed in MC-synthetase producing cyanobacteria, (i) small dots spread within the cytoplasm and (ii) densely packed cytoplasmic labeling, similar to the one of 16S rRNA probes (EUB338 and MICR3) from both Chrooccocale and Oscillatoriale species in cultures or in situ. Moreover, a focus on some M. aeruginosa cells from the PCC 7806 strain, cultivated in this study, shows a clear rRNA and mcyA-mRNA co-localized-labeling in the surrounding of densely packed plasmids (Figures 3A,B). McyA-mRNA labeled plasmids from PCC 7806 M. aeruginosa were also found extracellularly (Figure 3C, arrows). Previous TSA-FISH or DIG-FISH studies (Metcalf et al., 2009; Gan et al., 2010) on mcyA genes fluorescent detection have also shown some intracellular variability in accordance with toxin measurements. However, the lack of controls and information on time exposure during micrographs acquisition prevent from a clear understanding of presented micrographs.
The variations in mcyA-mRNA labeling observed in this study could be explained by the plasticity of microbial genome including that of toxic cyanobacteria (Figure 6) (Toussaint and Merlin, 2002; Tooming-Klunderud et al., 2008) where gene clusters are either integrated in chromosomal DNA with fewer mRNA transcripts, or can be multiplied in several hundreds of plasmids to rapidly synthetize metabolite through the production of intron-deprived mRNA (Schumann, 2008). We hypothesize that ribosomes targeted by MICR3 probe are either spread in the cytoplasm, or densely packed around the plasmids. This process could explain both the small dots in some Microcystis strains (Zeller et al., 2016) and Oscillatoriales (Figure 4D) and the co-localization of rRNA and mRNA Microcystis strains PCC7806 from this study (Figures 3A,B). Interestingly toxic strains have previously been shown to contain more extrachromosomal DNA than non-toxic ones (Schawbe et al., 1988), or to produce abundant mcyA PCR product (Valerio et al., 2009). All these results are coherent with the theory of horizontal gene transfer to explain sporadic distribution of mcy gene cluster in cyanobacteria (Kalaitzis et al., 2009). Figure 3C (arrows) also shows extracellular release of plasmids in M. aeruginosa, which is a well-known process in bacteria (Matsui et al., 2003) to respond to biotic or abiotic stresses (Paul and David, 1989; Alonso et al., 2000).
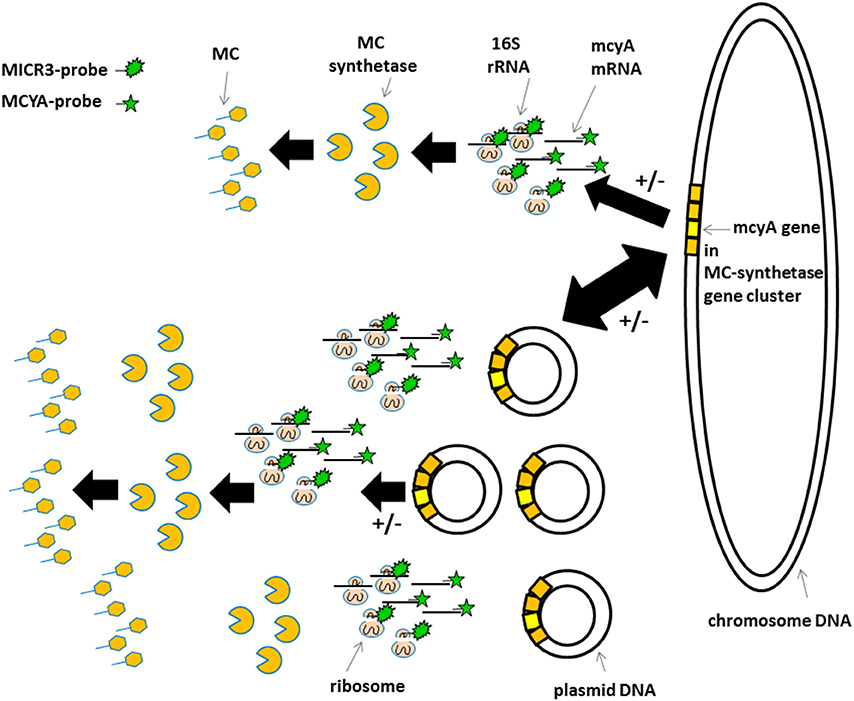
Figure 6. Schematic view of the proposed gene localization, transcription, and transduction steps involved in microcystin production. Top sequence in less toxic species and bottom sequence in very toxic species. +/− Indicate post-transcriptional and post-translational regulations mentioned by Schawbe et al. (1988), Valerio et al. (2009), and Kaebernick et al. (2000). Irregular green star show MICR3-probe targeting 16S rRNA. Regular green star show MCYA-probe targeting mcyA gene mRNA.
Conclusion
This study demonstrated that an easily accessible TSA-FISH protocol could be used as a diagnostic assay to characterize both the potential of a cyanobacterial bloom to be toxic by identifying Microcystis spp. with the specific MICR3 probe, and by simultaneously labeling all MC-producing species with MCYA probe. In addition, this powerful tool will help to further develop ecological and biomolecular studies on toxic cyanobacteria as visual localization of gene expression can be monitored.
Author Contributions
LB contributed to the different sections of this manuscript. NB aquired most TSA-FISH results. MP contributed to TSA-FISH results aquisition. MR did the specificity test and contributed to the results section. MB contributed to the manuscript. PZ, OP, and AM designed the FISH probes. IB designed the project and contributed to all sections of the manuscript.
Funding
We thank all partners of the FUI FISBOX project 2011–2015 MIO Marseille (N°F1105028U), University of Rennes 1, University of Paris Tech, Chrisar Society, Mermec Society, and Veolia and kindly acknowledges financial support from Minyvel Environment Society (France).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/article/10.3389/fenvs.2017.00043/full#supplementary-material
References
Alonso, M. C., Rodriguez, J., Rodriguez, J. J., and Borrego, J. J. (2000). Role of ciliates, flagellates and bacteriophages on the mortality of marine bacteria and on dissolved-DNA concentration in laboratory experimental systems. J. Exp. Mar. Biol. Ecol. 244, 239–252. doi: 10.1016/S0022-0981(99)00143-4
Amann, R., Binder, B. J., Olson, R. J., Chisholm, S. W., Devereaux, R., and Stahl, D. A. (1990). Combination of 16S ribosomal RNAtargeted oligonucleotide probes with flow cytometry for analyzing mixed microbial populations. Appl. Environ. Microbiol. 56, 1919–1925.
Biegala, I. C., Kenaway, G., and Alverca, E. (2002). Identification of bacteria associated with dinoflagellates (DINOPHYCEAE) Alexandrium spp. using tyramide signal amplification–fluorescent in situ hybridization and confocal microscopy. J. Phycol. 38, 404–411. doi: 10.1046/j.1529-8817.2002.01045.x
Biegala, I. C., Not, F., Vaulot, D., and Simon, N. (2003). Quantitative assessment of picoeukaryotes in the natural environment by using taxon-specific oligonucleotide probes in association with tyramide signal amplification-fluorescent in situ hybridization and flow cytometry. Appl. Environ. Microbiol. 69, 5519–5529. doi: 10.1128/AEM.69.9.5519-5529.2003
Biegala, I. C., and Raimbault, P. (2008). High abundance of diazotrophic picocyanobacteria (<3 μm) in a Southwest Pacific coral lagoon. Aquat. Microb. Ecol. 51, 45–53. doi: 10.3354/ame01185
Brient, L., Lengronne, M., Bertrand, E., Rolland, D., Sipel, A., Steinmann, D., et al. (2008). A phycocyanin probe as a tool for monitoring cyanobacteria in freshwater bodies. J. Environ. Monit. 10, 248–255. doi: 10.1039/B714238B
Carmichael, W. W., and An, J. S. (1999). Using an enzyme linked immunosorbent assay (ELISA) and a protein phosphatase inhibition assay (PPIA) for the detection of microcystins and nodularins. Nat. Toxins 7, 377–385. doi: 10.1002/1522-7189(199911/12)7:6<377::AID-NT80>3.0.CO;2-8
Chorus, I., and Bartram, J. (eds.). (1999). Toxic Cyanobacteria in Water. London; New York, NY: E & FN Spon, World Health Organization.
Gaget, V., Gribaldo, S., and Tandeau de Marsac, N. (2011). An rpoB signature sequence provides unique resolution for the molecular typing of cyanobacteria. Int. J. Syst. Evol. Microbiol. 61, 170–183. doi: 10.1099/ijs.0.019018-0
Gan, N., Huang, Q., Zheng, L., and Song, L. (2010). Quantitative assessement of toxic and nontoxic microcystis colonies in natural environments using fluorescence in situ hybridization and flow cytometry. Sci. China Life Sci. 53, 973–980. doi: 10.1007/s11427-010-4038-9
Hisbergues, M., Christiansen, G., Rouhiainen, L., Sivonen, K., and Borner, T. (2003). PCR-based identification of microcystin-producing genotypes of different cyanobacterial genera. Arch. Microbiol. 180, 402–410. doi: 10.1007/s00203-003-0605-9
Humpage, A. R., Froscio, S. M., Lau, H. M., Murphy, D., and Blackbeard, J. (2012). Evaluation of the Abraxis Strip Test for Microcystins™ for use with wastewater effluent and reservoir water. Water Res. 46, 1556–1565. doi: 10.1016/j.watres.2011.12.015
Kaebernick, M., Neilan, B. A., Borner, T., and Dittmann, E. (2000). Light and the transcriptional response of the microcystin biosynthesis gene cluster. Appl. Environ. Microbiol. 66, 3387–3392. doi: 10.1128/AEM.66.8.3387-3392.2000
Kalaitzis, J. A., Lauro, F. M., and Neilan, B. A. (2009). Mining cyanobacterial genomes for genes encoding complex biosynthetic pathways. Nat. Prod. Rep. 26 1447–1465. doi: 10.1039/b817074f
Lawton, L. A., and Edwards, C. (2008). “Conventional laboratory methods for cyanotoxins,” in Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs, Vol. 619. Advances in Experimental Medicine and Biology, ed H. K. Hudnell (New York, NY: Springer), 513–537.
Matsui, K., Ishii, N., and Kawabata, Z. (2003). Release of extracellular transformable plasmid DNA from Escherichia coli cocultivated with algae. Appl. Environ. Microbiol. 69, 2399–2404. doi: 10.1128/AEM.69.4.2399-2404.2003
Meissner, K., Dittmann, E., and Börner, T. (1996). Toxic and non-toxic strains of the cyanobacterium Microcystis aeruginosa contain sequences homologous to peptide synthetase genes. FEMS Microbiol. Lett. 135, 295–303. doi: 10.1111/j.1574-6968.1996.tb08004.x
Meriluoto, J., and Codd, G. A. (eds.). (2005). Cyanobacterial Monitoring and Cyanotoxin Analyses. Turku: Åbo Akademi University Press.
Metcalf, J. S., Reilly, M., Young, F. M., and Codd, G. A. (2009). Localization of microcystin synthetase genes in colonies of the cyanobacterium microcystis using fluorescence in situ hybridization1. J. Phycol. 45, 1400–1404. doi: 10.1111/j.1529-8817.2009.00750.x
Paul, J. H., and David, A. W. (1989). Production of extracellular nucleic acids by genetically altered bacteria in aquatic-environment microcosms. Appl. Environ. Microbiol. 55, 1865–1869.
Pearson, L. A., and Neilan, B. A. (2008). The molecular genetics of cyanobacterial toxicity as a basis for monitoring water quality and public health risk. Curr. Opin. Biotechnol. 19, 281–288. doi: 10.1016/j.copbio.2008.03.002
Pernthaler, A., and Amann, R. (2004). Simultaneous fluorescence in situ hybridization of mRNA and rRNA in environmental bacteria. Appl. Environ. Microbiol. 70, 5426–5433. doi: 10.1128/AEM.70.9.5426-5433.2004
Riou, V., Periot, M., and Biegala, I. C. (2017). Specific re-evaluation of oligonucleotide probes for detection of marine picoplankton by tyramide signal amplification-fluorescent in situ hybridization. Front. Microbiol. 8:854. doi: 10.3389/fmicb.2017.00854
Rudi, K., Skulberg, O. M., Skulberg, R., and Jakobsen, K. S. (2000). Application of sequence-specific labeled 16S rRNA gene oligonucleotide probes for genetic profiling of cyanobacterial abundance and diversity by array hybridization. Appl. Environ. Microbiol. 66, 4004–4011. doi: 10.1128/AEM.66.9.4004-4011.2000
Schawbe, W., Weihe, A., Börner, T., Henning, M., and Kohl, J. G. (1988). Plasmids in toxic and nontoxic strains of the cyanobacterium Microcystis aeruginosa. Curr. Microbiol. 17, 133–137. doi: 10.1007/BF01573468
Schonhuber, W., Zarda, B., Eix, S., Rippka, R., Herdman, M., Ludwig, W., et al. (1999). In situ identification of cyanobacteria with horseradish peroxidase-labeled, rRNA-targeted oligonucleotide probes. Appl. Environ. Microbiol. 65, 1259–1267.
Schumann, W. (2008). “Escherichia coli cloning and expression vectors,” in Plasmids: Current Research and Future Trends, ed G. Lipps (Caister: Academic Press), 1–2.
Shirai, M., Matsumaru, K., Ohotake, A., Takamura, Y., Aida, T., and Nakano, M. (1989). Development of a solid medium for growth and isolation of axenic Microcystis strains. Appl. Environ. Microbiol. 55, 2569–2571.
Shirai, M., Ohtake, A., Sano, T., Matsumoto, S., Sakamoto, T., Sato, A., et al. (1991). Toxicity and toxins of natural blooms and isolated strains of Microcystis spp. (Cyanobacteria) and improved procedure for purification of cultures. Appl. Environ. Microbiol. 57, 1241–1245.
Spoof, L., Vesterkvist, P., Lindholm, T., and Meriluoto, J. (2003). Screening for cyanobacterial hepatotoxins, microcystins and nodularin in environmental water samples by reversed-phase liquid chromatography-electrospray ionization mass spectrometry. J. Chromatogr. A 1020, 105−119. doi: 10.1016/S0021-9673(03)00428-X
Tillett, D., Dittmann, E., Erhard, M., von Döhren, H., Börner, T., and Neilan, B. A. (2000). Structural organization of microcystin biosynthesis in Microcystis aeruginosa PCC7806: an integrated peptide-polyketide synthetase system. Chem. Biol. 7, 753–764. doi: 10.1016/s1074-5521(00)00021-1
Tooming-Klunderud, A., Mikalsen, B., Kristensen, K., and Jakobsen, K. S. (2008). The mosaic structure of the mcyABC operon in Microcystis. Microbiology 154, 1886–1899. doi: 10.1099/mic.0.2007/015875-0
Toussaint, A., and Merlin, C. (2002). Mobile elements as a combination of functional modules. Plasmid 47, 26–35. doi: 10.1006/plas.2001.155
Trcek, T., Chao, J. A., Larson, D. R., Park, H. Y., Zenklusen, D., Shenoy, S. M., et al. (2012). Single-mRNA counting using fluorescent in situ hybridization in budding yeast. Nat. Protoc. 7, 408–419. doi: 10.1038/nprot.2011.451
Urdea, M. S., Warner, B. D., Running, J. A., Stempien, M., Clyne, J., and Horn, T. (1988). A comparison of non-radioisotopic hybridization assay methods usingfluorescent, chemiluminescent and enzyme labeled synthetic oligode-oxyribonucleotide probes. Nucleic Acids Res. 16, 4937–4956. doi: 10.1093/nar/16.11.4937
Valerio, E., Chambel, L., Paulino, S., Faria, N., Pereira, P., and Tenreiro, R. (2009). Molecular identification, typing and traceability of cyanobacteria from freshwater reservoirs. Microbiology 155, 642–656. doi: 10.1099/mic.0.022848-0
West, N. J., Schönuber, W. A., Fuller, N., Amann, R., Rippka, R., Post, A., et al. (2001). Closely related Prochlorococcus genotypes show remarkably different depth distributions in two oceanic regions as revealed by in situ hybridization using 16S rRNA-targetes oligonucleotides. Microbiology 147, 1731–1744. doi: 10.1099/00221287-147-7-1731
Keywords: microcystin, cyanobacteria toxin, freshwater, FISH, epifluorescence, mRNA, TSA-FISH, CARD-FISH
Citation: Brient L, Ben Gamra N, Periot M, Roumagnac M, Zeller P, Bormans M, Méjean A, Ploux O and Biegala IC (2017) Rapid Characterization of Microcystin-Producing Cyanobacteria in Freshwater Lakes by TSA-FISH (Tyramid Signal Amplification-Fluorescent In Situ Hybridization). Front. Environ. Sci. 5:43. doi: 10.3389/fenvs.2017.00043
Received: 07 April 2017; Accepted: 27 June 2017;
Published: 25 July 2017.
Edited by:
Rajeshwar P. Sinha, Banaras Hindu University, IndiaReviewed by:
Pawan K. Dadheech, Central University of Rajasthan, IndiaRadha Prasanna, Indian Agricultural Research Institute, India
Copyright © 2017 Brient, Ben Gamra, Periot, Roumagnac, Zeller, Bormans, Méjean, Ploux and Biegala. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Luc Brient, luc.brient@univ-rennes1.fr
Isabelle C. Biegala, isabelle.biegala@ird.fr
 Luc Brient
Luc Brient Nihel Ben Gamra
Nihel Ben Gamra Marine Periot
Marine Periot Marie Roumagnac2
Marie Roumagnac2  Myriam Bormans
Myriam Bormans Olivier Ploux
Olivier Ploux Isabelle C. Biegala
Isabelle C. Biegala